Understanding Infertility Terminology: Your Complete Fertility Glossary

 Navigating the world of fertility treatments can feel overwhelming. Medical professionals use specialized language that sometimes sounds like a foreign dialect. When you hear terms like intracytoplasmic sperm injection or assisted reproductive technology for the first time, confusion is natural. This comprehensive infertility terminology guide breaks down complex medical vocabulary into clear, understandable language.
Navigating the world of fertility treatments can feel overwhelming. Medical professionals use specialized language that sometimes sounds like a foreign dialect. When you hear terms like intracytoplasmic sperm injection or assisted reproductive technology for the first time, confusion is natural. This comprehensive infertility terminology guide breaks down complex medical vocabulary into clear, understandable language.
Understanding fertility-related terms empowers you to make informed decisions about your reproductive health. Whether you’re just beginning to explore fertility options or you’re already in treatment, knowing what these words mean helps you communicate effectively with your healthcare team. This glossary covers everything from basic reproductive anatomy to advanced assisted reproductive procedures.
Medical terminology should never stand between you and understanding your treatment path. Each definition in this guide is written with clarity in mind. You’ll find explanations for common fertility terms, IVF vocabulary, and reproductive health language that healthcare providers use daily. Knowledge transforms anxiety into confidence.
Questions About These Terms?
Our fertility specialists understand that medical terminology can be confusing. We’re here to explain your specific situation in plain language and answer all your questions.
Planning surrogacy abroad?
Read our complete Guide to International Surrogacy to understand the process, costs, legal rules, and timeline for international intended parents.
Additional Resources to Read:
Surrogacy Services in Georgia Country
Understanding Infertility: Core Concepts and Definitions
Infertility refers to the inability to conceive after one year of regular unprotected intercourse for women under 35, or six months for women over 35. This medical condition affects approximately one in eight couples worldwide. The term encompasses various underlying causes that prevent pregnancy from occurring naturally. Both male and female factors contribute to infertility in roughly equal proportions.
Primary vs. Secondary Infertility
Primary infertility
 Navigating the world of fertility treatments can feel overwhelming. Medical professionals use specialized language that sometimes sounds like a foreign dialect. When you hear terms like intracytoplasmic sperm injection or assisted reproductive technology for the first time, confusion is natural. This comprehensive infertility terminology guide breaks down complex medical vocabulary into clear, understandable language.
Navigating the world of fertility treatments can feel overwhelming. Medical professionals use specialized language that sometimes sounds like a foreign dialect. When you hear terms like intracytoplasmic sperm injection or assisted reproductive technology for the first time, confusion is natural. This comprehensive infertility terminology guide breaks down complex medical vocabulary into clear, understandable language.
Understanding fertility-related terms empowers you to make informed decisions about your reproductive health. Whether you’re just beginning to explore fertility options or you’re already in treatment, knowing what these words mean helps you communicate effectively with your healthcare team. This glossary covers everything from basic reproductive anatomy to advanced assisted reproductive procedures.
Medical terminology should never stand between you and understanding your treatment path. Each definition in this guide is written with clarity in mind. You’ll find explanations for common fertility terms, IVF vocabulary, and reproductive health language that healthcare providers use daily. Knowledge transforms anxiety into confidence.
Questions About These Terms?
Our fertility specialists understand that medical terminology can be confusing. We’re here to explain your specific situation in plain language and answer all your questions.
Planning surrogacy abroad?
Read our complete Guide to International Surrogacy to understand the process, costs, legal rules, and timeline for international intended parents.
Additional Resources to Read:
Surrogacy Services in Georgia Country
Understanding Infertility: Core Concepts and Definitions
Infertility refers to the inability to conceive after one year of regular unprotected intercourse for women under 35, or six months for women over 35. This medical condition affects approximately one in eight couples worldwide. The term encompasses various underlying causes that prevent pregnancy from occurring naturally. Both male and female factors contribute to infertility in roughly equal proportions.
Primary vs. Secondary Infertility
Primary infertility describes couples who have never achieved pregnancy despite trying. These individuals have no history of conception. Secondary infertility occurs when couples have conceived before but cannot achieve another pregnancy. Both types present unique emotional and medical challenges requiring different approaches to treatment and support.
Male Factor Infertility
Male infertility accounts for about 40% of all infertility cases. Issues with sperm production, sperm quality, or sperm delivery create challenges for conception. Sperm count refers to the number of sperm cells present in a semen sample. Low sperm count means fewer than 15 million sperm per milliliter of semen. Sperm motility describes how well sperm move and swim toward an egg.
Motile sperm are those capable of forward movement necessary to reach and fertilize an egg. Poor sperm motility reduces the chances of natural conception. Sperm morphology examines the shape and structure of sperm cells. Abnormally shaped sperm may struggle to penetrate an egg successfully. These three factors combine to determine overall male fertility potential.
Female Factor Infertility
Female infertility involves issues with the reproductive tract, hormonal imbalances, or egg quality problems. The fallopian tubes transport eggs from ovaries to the uterus. Blocked or damaged fallopian tubes prevent sperm from reaching eggs. Ovulation disorders represent another common cause. When ovaries don’t release eggs regularly, conception becomes difficult or impossible.
The uterus must provide a healthy environment for embryo implantation. Structural problems or uterine lining issues interfere with this process. Age significantly impacts female fertility through declining egg quantity and quality. These factors often work in combination rather than isolation.
Confused about whether your situation involves male or female factors? Call +91-8800481100 to discuss your specific circumstances with our fertility experts.
Assisted Reproductive Technology (ART): Comprehensive Overview
Assisted reproductive technology encompasses all fertility treatments where both eggs and sperm are handled outside the body. These procedures help couples overcome various infertility obstacles. ART represents modern medicine’s most advanced approach to treating reproductive challenges. Success rates vary based on age, diagnosis, and specific procedure type.
What ART Includes
The assisted reproductive technology category includes several distinct procedures. In vitro fertilization forms the foundation of most ART treatments. Intracytoplasmic sperm injection represents a specialized technique within IVF. Egg and embryo cryopreservation allow patients to preserve fertility for future use. Donor egg and donor sperm programs help patients who cannot use their own genetic material.
Gestational surrogacy involves another woman carrying a pregnancy. Preimplantation genetic testing screens embryos for genetic conditions. Each ART procedure addresses specific fertility challenges. Some patients require multiple procedures combined to achieve pregnancy.
ART Success Factors
Multiple factors influence ART outcomes. Patient age plays the most significant role in success rates. Women under 35 typically experience higher success rates than older patients. The cause of infertility affects treatment effectiveness. Previous pregnancy history provides insight into potential outcomes. Embryo quality directly impacts implantation chances.
The number of embryos transferred influences both success rates and multiple pregnancy risks. Clinic experience and laboratory quality matter enormously. Proper patient selection and protocol customization improve results. Lifestyle factors including weight, smoking, and alcohol consumption affect outcomes.
Understanding Live Birth Rates
Live birth rates represent the ultimate measure of ART success. This statistic counts actual babies born, not just positive pregnancy tests. One cycle of treatment may or may not result in a live birth. Multiple cycles often prove necessary to achieve the desired outcome. National registries track live birth statistics to help patients understand realistic expectations.
Age-specific live birth rates provide the most relevant information for individual patients. Cumulative live birth rates across multiple cycles offer a more complete picture. Understanding these statistics helps patients set realistic expectations and make informed decisions about treatment continuation.
Ready to Explore ART Options?
Our team specializes in personalized assisted reproductive technology treatment plans. We’ll help you understand which procedures offer the best chance of success for your unique situation.
In Vitro Fertilization (IVF) Terminology Explained
In vitro fertilization literally means fertilization in glass. This process occurs in a laboratory dish rather than inside a woman’s body. IVF has helped millions of couples worldwide achieve pregnancy since the first successful procedure in 1978. The technique involves multiple steps that span several weeks from start to finish.
The IVF Cycle Process
An IVF cycle begins with ovarian stimulation. Women take hormone medications to encourage multiple eggs to develop simultaneously. Normally, ovaries release one egg per month. Stimulation increases this number significantly. More eggs provide more opportunities for successful fertilization and embryo development.
Monitoring occurs throughout stimulation through ultrasound examinations and blood tests. These procedures track follicle growth and hormone levels. When eggs reach maturity, a trigger shot initiates final egg maturation. Egg retrieval takes place approximately 36 hours after the trigger injection. This minor surgical procedure collects eggs from the ovaries using ultrasound guidance.
Fertilization and Embryo Development
After retrieval, embryologists combine eggs with sperm in the laboratory. Conventional IVF places sperm and eggs together, allowing natural fertilization to occur. Intracytoplasmic sperm injection involves directly injecting a single sperm into each egg. This technique helps when sperm quality or quantity issues exist.
Fertilized eggs become embryos. Embryologists observe embryo development over several days. Day 3 embryos typically contain six to eight cells. Blastocyst embryos develop by day 5 or 6, containing over 100 cells. Many clinics prefer transferring blastocysts because they better predict successful implantation.
Embryo Transfer and Implantation
Embryo transfer places one or more embryos into the uterus. This simple procedure requires no anesthesia and resembles a pap smear. A thin catheter carries embryos through the cervix into the uterine cavity. Ultrasound guidance ensures proper placement. The entire process takes just minutes to complete.
Implantation occurs when an embryo attaches to the uterine lining. This natural process happens several days after transfer. Not all transferred embryos successfully implant. The two-week wait between transfer and pregnancy testing creates significant emotional stress for patients. Blood tests measure hormone levels to confirm pregnancy.
Luteal Phase Support
The luteal phase occurs after ovulation or egg retrieval. Progesterone hormone prepares the uterine lining for implantation. IVF patients typically require progesterone supplementation because retrieval removes cells that normally produce this hormone. Support continues through early pregnancy if conception occurs. Various progesterone formulations include injections, vaginal suppositories, and oral medications.
Want to understand how IVF could work for you specifically? Our specialists explain every step in detail. Call +91-8800481100 for a personalized consultation.
Intracytoplasmic Sperm Injection and Specialized Fertility Procedures
Intracytoplasmic sperm injection revolutionized treatment for severe male infertility. This precise procedure involves injecting a single sperm directly into an egg’s cytoplasm. ICSI bypasses many natural barriers that prevent fertilization. The technique requires highly skilled embryologists and specialized equipment. Success rates often match or exceed conventional IVF fertilization methods.
When ICSI Is Recommended
Several conditions indicate ICSI as the preferred fertilization method. Very low sperm count makes conventional fertilization unlikely to succeed. Poor sperm motility prevents sperm from reaching eggs naturally. Abnormal sperm morphology reduces penetration capability. Previous IVF cycles with failed fertilization suggest ICSI for subsequent attempts.
Using frozen sperm often requires ICSI because freezing may damage some sperm cells. Surgical sperm extraction from the testes always necessitates ICSI. Eggs with thick outer shells may benefit from direct injection. Some clinics recommend ICSI for all patients to maximize fertilization rates.
The ICSI Procedure Step by Step
ICSI begins with careful sperm selection. Embryologists choose the healthiest-appearing sperm under high magnification. Individual sperm selection considers morphology and motility. The selected sperm is drawn into a tiny glass needle. Simultaneously, an egg is held steady with a holding pipette.
The injection needle pierces the egg membrane carefully. The sperm is deposited directly into the egg’s interior. This process repeats for each mature egg. After injection, eggs are cultured in specialized media. Fertilization confirmation occurs the following day when two pronuclei appear inside the egg.
Advanced Sperm Selection Techniques
Several advanced techniques help identify the best sperm for ICSI. Physiological intracytoplasmic sperm injection uses hyaluronic acid to select mature sperm. This substance mimics the egg’s natural coating. Only mature sperm bind to hyaluronic acid, improving selection accuracy. The technique may reduce miscarriage rates and increase pregnancy success.
Intracellular sperm morphology assessment examines sperm at extremely high magnification. This allows embryologists to detect subtle structural abnormalities invisible under standard magnification. Selecting sperm with normal internal structures may improve outcomes. Research continues regarding the effectiveness of these enhanced selection methods.
Surgical Sperm Retrieval
Some men produce sperm but cannot ejaculate them naturally. Surgical procedures extract sperm directly from the reproductive tract. Testicular sperm extraction removes tissue samples from the testes. Percutaneous epididymal sperm aspiration uses a needle to withdraw sperm from the epididymis. Microsurgical epididymal sperm aspiration uses surgical magnification for precise sperm collection.
These procedures help men with obstructive azoospermia achieve biological fatherhood. Non-obstructive azoospermia occurs when sperm production is severely reduced. Microdissection testicular sperm extraction can sometimes find small pockets of sperm production. All surgically retrieved sperm require ICSI for fertilization.
Is ICSI Right for Your Situation?
Male factor infertility doesn’t mean you can’t have biological children. Our specialists evaluate your specific case and recommend the most effective fertilization approach.
Understanding Eggs, Embryos, and Early Development
The oocyte is the medical term for an egg cell. Women are born with approximately one to two million oocytes. This number decreases throughout life. By puberty, about 300,000 remain. Each month during reproductive years, one oocyte typically matures and releases during ovulation. Only about 300 to 400 eggs ever ovulate during a woman’s lifetime.
Egg Maturity and Quality
Not all retrieved eggs are mature enough for fertilization. Germinal vesicle eggs are immature and cannot be fertilized immediately. Metaphase I eggs have started maturation but haven’t completed the process. Metaphase II eggs are fully mature and ready for fertilization. Only metaphase II eggs can be used for IVF or ICSI procedures.
Egg quality significantly impacts fertilization success and embryo development. Age affects egg quality more than any other factor. Chromosomal abnormalities increase in eggs as women age. These abnormalities reduce fertilization rates, increase miscarriage risk, and lower live birth chances. No current treatment can reverse age-related egg quality decline.
Fertilization and Early Embryo Development
Fertilization occurs when sperm penetrates the egg. The fertilized egg is now called a zygote. Cell division begins within hours of fertilization. The two-cell embryo appears approximately 24 hours after fertilization. Division continues rapidly over subsequent days. Four-cell embryos develop around day two. Eight-cell embryos form by day three.
At this stage, embryos are also called cleavage-stage embryos. Each cell is called a blastomere. The embryo continues dividing while traveling toward the uterus in natural conception. In IVF, embryos remain in culture dishes during this time. Embryologists monitor development daily, documenting progress and quality.
Morula and Blastocyst Formation
Around day four, the embryo becomes a morula. This solid ball of cells contains 16 to 32 blastomeres. The cells begin to differentiate and organize. A cavity forms inside the embryo. When this cavity appears, the embryo becomes a blastocyst. This typically occurs on day five or six after fertilization.
Blastocysts contain two distinct cell types. The inner cell mass will become the fetus. The trophectoderm cells will form the placenta and membranes. Blastocyst quality grading assesses expansion, inner cell mass, and trophectoderm. Higher-grade blastocysts have better implantation potential. Many clinics prefer transferring blastocysts rather than earlier-stage embryos.
Embryo Grading Systems
Embryologists use standardized grading systems to evaluate embryo quality. Day-three embryos are graded based on cell number, cell symmetry, and fragmentation percentage. Ideal day-three embryos have seven to nine cells with minimal fragmentation. Even cell sizes indicate better developmental potential.
Blastocyst grading considers three factors. Expansion describes how developed the cavity is. Inner cell mass quality predicts fetal development potential. Trophectoderm quality indicates placental development capability. A blastocyst might be graded as 4AA, with the number representing expansion and letters representing inner cell mass and trophectoderm quality respectively.
Questions about egg or embryo quality in your specific case? Our embryologists can explain your results in understandable terms. Call +91-8800481100 today.
Fertility Hormones and Ovarian Stimulation Protocols
Hormones control the entire reproductive process. Understanding these chemical messengers helps explain fertility treatments. Follicle-stimulating hormone prompts follicles in the ovaries to grow. Each follicle contains one egg. FSH levels naturally rise at the beginning of each menstrual cycle. Injectable FSH medications stimulate multiple follicles simultaneously during IVF.
Key Reproductive Hormones
Luteinizing hormone triggers ovulation in natural cycles. The LH surge signals an egg to complete maturation and release. IVF uses injectable human chorionic gonadotropin as a trigger shot because it mimics LH. This hormone also supports early pregnancy. Pregnancy tests detect hCG in urine or blood.
Estrogen rises as follicles grow. This hormone thickens the uterine lining and prepares it for implantation. High estrogen levels during stimulation indicate good follicle development. Progesterone prepares and maintains the uterine lining after ovulation. It prevents menstruation if pregnancy occurs. All IVF patients need progesterone supplementation after egg retrieval.
Anti-Müllerian hormone helps predict ovarian reserve. Higher AMH levels suggest more remaining eggs. This hormone helps doctors determine appropriate medication dosing. Thyroid hormones affect overall reproductive function. Thyroid disorders require correction before starting fertility treatment.
Ovarian Stimulation Medications
Multiple protocols exist for ovarian stimulation. Gonadotropins are injectable hormones that stimulate follicle development. These medications contain FSH alone or FSH combined with LH. Daily injections continue for approximately 8 to 14 days. Doctors adjust doses based on ultrasound and blood test results.
GnRH antagonists prevent premature ovulation during stimulation. These daily injections typically start when follicles reach a certain size. GnRH agonists can serve similar functions but work differently. Long protocols use agonists to suppress natural hormones before stimulation begins. Short protocols start agonists during stimulation.
Oral medications sometimes supplement injection protocols. Clomiphene citrate stimulates natural FSH production. Letrozole also promotes follicle development through a different mechanism. These drugs cost less than injectables but generally produce fewer eggs. Some mild stimulation protocols combine oral medications with low-dose injectables.
Monitoring During Stimulation
Patients undergo frequent monitoring throughout stimulation. Transvaginal ultrasound examinations measure follicle size and count. Technicians measure each visible follicle on the ovaries. Mature follicles typically measure 16 to 22 millimeters in diameter. Blood tests measure estrogen levels to confirm follicle maturity.
Rising estrogen indicates growing follicles. Very high estrogen levels may indicate ovarian hyperstimulation risk. Monitoring continues every few days initially, then daily as follicles mature. This information guides medication adjustments and determines trigger shot timing. Precise timing ensures optimal egg maturity at retrieval.
Trigger Shot and Final Maturation
The trigger shot initiates final egg maturation. This injection must occur at precisely the right time. Too early means eggs won’t be mature enough. Too late risks ovulation before retrieval. Most triggers use human chorionic gonadotropin. Some protocols use a GnRH agonist as a trigger instead.
Egg retrieval is scheduled exactly 35 to 36 hours after the trigger injection. This timing allows eggs to complete maturation without releasing from follicles. Patients must follow trigger timing instructions precisely. Even a few hours difference can significantly impact results. Some protocols use a dual trigger combining both hCG and GnRH agonist.
Concerned About Medication Protocols?
Every stimulation protocol is customized to your unique situation. Our specialists explain medication purposes, side effects, and what to expect throughout treatment.
Diagnostic Procedures and Fertility Testing Terminology
Accurate diagnosis forms the foundation of effective fertility treatment. Multiple tests evaluate both male and female fertility factors. Results guide treatment selection and predict success probabilities. Most fertility workups begin with simple, non-invasive tests before progressing to more complex procedures.
Semen Analysis Basics
Semen analysis provides essential information about male fertility potential. This test evaluates several key parameters. Volume measures the amount of ejaculated semen. Normal volume ranges from 1.5 to 5 milliliters. Low volume may indicate collection problems or reproductive tract issues. Very high volume can dilute sperm concentration.
Sperm concentration counts sperm cells per milliliter. Normal concentration exceeds 15 million per milliliter. Total sperm count multiplies concentration by volume. At least 39 million total sperm should be present. Oligospermia describes low sperm count. Azoospermia means no sperm are present in the ejaculate.
Progressive motility measures forward-moving sperm. At least 32% of sperm should show progressive movement. Total motility includes all moving sperm. Normal morphology examines sperm shape. At least 4% should have normal form by strict criteria. Vitality testing determines the percentage of live sperm. Additional tests may examine DNA fragmentation or antibody presence.
Female Hormone Testing
Baseline hormone testing occurs early in the menstrual cycle. Day two or three blood tests measure FSH, LH, and estradiol. These values help assess ovarian reserve and function. Elevated FSH suggests diminished ovarian reserve. High LH relative to FSH may indicate polycystic ovary syndrome. Low estradiol combined with high FSH confirms poor ovarian function.
Anti-Müllerian hormone testing can occur any cycle day. This value remains relatively stable throughout the cycle. Higher AMH suggests better ovarian reserve. Very low AMH indicates few remaining eggs. Very high AMH may indicate PCOS. Progesterone testing confirms ovulation occurrence. This test happens approximately one week after suspected ovulation.
Thyroid function tests evaluate TSH and sometimes additional thyroid hormones. Abnormal thyroid function interferes with conception and pregnancy maintenance. Prolactin elevation can prevent ovulation. This hormone requires evaluation if menstrual irregularity exists. Additional hormone tests may be ordered based on symptoms and initial results.
Ultrasound Examinations
Transvaginal ultrasound provides detailed views of reproductive organs. This procedure uses a probe inserted into the vagina. Sound waves create images of the uterus and ovaries. Antral follicle count occurs during early cycle baseline ultrasounds. Small follicles visible on the ovaries predict how many eggs might develop during stimulation.
Ultrasound evaluates uterine structure and identifies abnormalities. Fibroids appear as solid masses in or around the uterus. Polyps project into the uterine cavity. Both conditions may interfere with implantation. Ultrasound measures uterine lining thickness. Adequate thickness is necessary for embryo implantation. Ovarian cysts or masses are also visible on ultrasound.
Hysterosalpingography and Tubal Assessment
Hysterosalpingography evaluates fallopian tube patency. This X-ray procedure involves injecting contrast dye through the cervix. The dye fills the uterine cavity and flows through fallopian tubes. X-rays capture images as dye progresses. Open tubes allow dye to spill into the abdominal cavity. Blocked tubes prevent dye passage.
This test causes some discomfort but typically requires no anesthesia. Cramping occurs as dye distends the uterus. The procedure also reveals uterine shape abnormalities. Saline infusion sonography serves as an alternative tube-testing method. Ultrasound visualization occurs as sterile saline fills the uterus. Bubbles visible in the tubes suggest patency.
Advanced Diagnostic Procedures
Hysteroscopy allows direct visualization of the uterine cavity. A thin telescope passes through the cervix. Doctors can diagnose and sometimes treat abnormalities during the same procedure. Small polyps or fibroids can be removed. Uterine septum or scar tissue can be corrected. Hysteroscopy requires anesthesia and occurs in a surgical setting.
Laparoscopy examines the pelvic cavity. Small incisions allow camera insertion. Surgeons can evaluate endometriosis, adhesions, and tubal damage. Treatment often occurs simultaneously with diagnosis. This procedure requires general anesthesia and brief recovery time. Endometrial biopsy samples uterine lining tissue. Analysis can reveal inflammation, infection, or timing issues.
Not sure which tests you need? Our fertility specialists create comprehensive diagnostic plans tailored to your situation. Call +91-8800481100 to schedule your evaluation.
Embryo Transfer Procedures and Implantation Process
Embryo transfer represents the final step in an IVF cycle. This relatively simple procedure determines whether treatment succeeds or fails. Despite its simplicity, technique matters significantly. Proper placement increases implantation chances. The procedure causes minimal discomfort and requires no anesthesia in most cases.
Transfer Day Preparation
Patients arrive with a moderately full bladder. Fluid in the bladder helps visualize the uterus via abdominal ultrasound. Too much fluid causes discomfort. Too little prevents adequate visualization. Timing the perfect bladder fullness requires practice. Some patients need to partially empty their bladder before the procedure.
The embryologist prepares selected embryos for transfer. Embryos are loaded into a thin, flexible catheter. This occurs in the laboratory moments before transfer. The physician confirms embryo presence in the catheter. Mock transfers may have occurred previously to plan the exact approach. Cervical preparation with gentle cleaning occurs before catheter insertion.
The Transfer Procedure
The patient lies in a position similar to a pap smear. A speculum allows cervical visualization. The physician gently passes the catheter through the cervix into the uterus. Abdominal ultrasound guidance ensures correct placement. The ideal location is in the upper-middle portion of the uterine cavity. The catheter should not touch the fundus or upper uterine wall.
When proper position is confirmed, embryos are gently expelled. The catheter remains still during expulsion to avoid dislodging embryos. After transfer, the catheter is carefully removed. The embryologist immediately examines the catheter under a microscope. This confirms all embryos left the catheter successfully. Occasionally, an embryo adheres to the catheter and requires a second transfer attempt.
Fresh Versus Frozen Embryo Transfer
Fresh embryo transfer occurs three to six days after egg retrieval. The patient’s body is still recovering from stimulation. Hormone levels remain elevated from trigger medications. Some evidence suggests frozen embryo transfer may achieve higher success rates. The uterus may be more receptive when not recently stimulated.
Frozen embryo transfer occurs in a subsequent cycle. All embryos undergo cryopreservation after development. The uterus is prepared using hormone medications or timed with natural ovulation. This approach allows time for recovery from stimulation. Genetic testing can be completed before transfer. Remaining embryos are available for future attempts without repeating stimulation and retrieval.
Single Versus Multiple Embryo Transfer
Single embryo transfer means placing one embryo in the uterus. This approach minimizes multiple pregnancy risk. Twin and higher-order pregnancies carry increased complications. Mother and babies face higher risks than singleton pregnancies. Many clinics now advocate for elective single embryo transfer, especially for younger patients with high-quality embryos.
Multiple embryo transfer increases pregnancy chances per cycle. However, it also dramatically increases multiple pregnancy risk. Two embryos might both implant. One embryo might split into identical twins. The result could be triplets from transferring two embryos. Pregnancy and delivery complications rise significantly with multiples. Individual circumstances guide the decision about embryo number.
Understanding Implantation
Implantation begins several days after transfer. The blastocyst hatches from its protective shell. Trophectoderm cells begin invading the uterine lining. This process triggers local immune and vascular changes. Blood vessels form connections between the embryo and maternal circulation. The developing placenta secretes human chorionic gonadotropin hormone.
Successful implantation depends on embryo quality and uterine receptivity. Both must be optimal at the same time. The implantation window refers to the brief period when the uterus is receptive. This typically occurs six to ten days after ovulation. Timing embryo transfer to coincide with this window maximizes success. Endometrial receptivity testing can identify individual window timing.
Questions About Your Upcoming Transfer?
Embryo transfer is a crucial moment in your fertility journey. Our team ensures you understand every aspect of the procedure and what to expect during the waiting period afterward.
Cryopreservation and Fertility Preservation Options
Cryopreservation freezes biological materials for future use. Fertility preservation allows people to maintain reproductive options. Cancer patients often preserve fertility before treatment begins. People delaying parenthood can freeze eggs or embryos. Same-sex couples and single individuals utilize preservation as part of family planning. The technology has improved dramatically over recent decades.
Egg Freezing (Oocyte Cryopreservation)
Egg freezing preserves unfertilized eggs. Women undergo ovarian stimulation identical to IVF. Egg retrieval collects mature eggs. Embryologists immediately freeze eggs using vitrification. This ultra-rapid freezing prevents ice crystal formation. Ice crystals would damage delicate egg structures. Vitrification transformed egg freezing from experimental to routine.
Frozen eggs can remain in storage for many years. No definitive time limit exists for storage. Eggs thawed years later show similar survival and fertilization rates. Success depends heavily on age at freezing. Younger eggs yield better outcomes than eggs frozen at older ages. Women typically freeze 15 to 20 eggs for optimal pregnancy chances later.
When ready to use frozen eggs, they undergo thawing. Survival rates exceed 90% with vitrification. Surviving eggs are fertilized using ICSI. This injection technique prevents damage from sperm binding attempts. Embryos develop normally and are transferred or refrozen. Pregnancy rates from frozen eggs approach those from fresh eggs when age-matched.
Embryo Freezing (Embryo Cryopreservation)
Embryo cryopreservation freezes fertilized embryos. Couples often create more embryos than can be safely transferred. Excess embryos are frozen for future use. This avoids repeating stimulation and egg retrieval for subsequent pregnancies. Embryo freezing has been performed successfully since the 1980s. Modern vitrification techniques yield excellent survival rates.
Embryos can be frozen at various developmental stages. Day-three embryos and blastocysts both freeze well. Many clinics prefer freezing blastocysts because they tolerate the process well. Survival rates often exceed 95%. Frozen embryo transfer cycles require endometrial preparation. The uterine lining must be ready to receive the embryo.
Patients taking ownership of frozen embryos face important decisions. Embryos can be stored indefinitely while paying storage fees. They can be used for future pregnancies. Donation to other couples helps those unable to create embryos. Donation to research contributes to scientific advancement. Some choose to discontinue storage and allow thawing without use.
Sperm Freezing (Sperm Cryopreservation)
Sperm freezing preserves male fertility. Men facing cancer treatment routinely freeze sperm beforehand. Chemotherapy and radiation often damage sperm production. Recovery may take years or never occur. Frozen sperm provides insurance against this risk. Men with declining sperm parameters may bank sperm while quality remains adequate.
The freezing process involves mixing sperm with cryoprotectant. Samples are divided into multiple vials. This allows using one vial per IVF attempt. Slow-rate freezing or vitrification methods both work for sperm. Sperm tolerate freezing better than eggs. Survival rates typically remain high. Frozen sperm can be used for IUI, IVF, or ICSI procedures.
Surgically retrieved sperm are always frozen initially. Recovery typically yields limited quantities. Using fresh surgical sperm would require perfect IVF cycle timing. Freezing allows flexible scheduling. Men undergoing vasectomy might freeze sperm beforehand. This maintains options if life circumstances change later.
Ovarian Tissue Freezing
Ovarian tissue cryopreservation represents the newest preservation option. This technique helps women who cannot delay cancer treatment for ovarian stimulation. Surgery removes all or part of one ovary. The outer cortex containing thousands of immature follicles is frozen. After cancer recovery, tissue can be transplanted back. This has resulted in natural pregnancies and live births.
The procedure remains experimental but shows promise. Very young girls can preserve fertility before puberty. Mature eggs cannot be retrieved before puberty begins. Ovarian tissue offers their only current preservation option. Future technologies may allow maturing frozen follicles in the laboratory. Research continues to improve outcomes with this innovative approach.
Considering fertility preservation? Time matters, especially before medical treatments. Call +91-8800481100 to discuss your options and timing.
Preimplantation Genetic Testing and Screening
Preimplantation genetic testing examines embryos before transfer. This technology identifies chromosomal abnormalities and genetic conditions. Testing helps select the healthiest embryos. Several types of genetic testing serve different purposes. All require embryo biopsy to obtain cells for analysis. Results guide embryo selection decisions.
Preimplantation Genetic Testing for Aneuploidies
Preimplantation genetic testing for aneuploidies screens chromosome numbers. Humans normally have 23 pairs of chromosomes. Aneuploid embryos have extra or missing chromosomes. Most aneuploidies prevent implantation or cause early miscarriage. Down syndrome results from an extra chromosome 21. Monosomy X causes Turner syndrome. Trisomy 13 and 18 are usually fatal.
Aneuploidy increases dramatically with maternal age. Women under 35 produce about 50% aneuploid embryos. This percentage exceeds 75% by age 40. Testing identifies chromosomally normal embryos for transfer. This may reduce miscarriage rates and improve live birth rates per transfer. However, testing doesn’t improve overall cumulative success rates across multiple cycles.
The biopsy typically occurs at the blastocyst stage. Several trophectoderm cells are removed. These will become placenta, not baby. The embryo is frozen while awaiting results. Testing usually takes one to two weeks. Normal embryos are transferred in a subsequent cycle. Mosaic embryos contain both normal and abnormal cells. Management of mosaic results requires careful counseling.
Preimplantation Genetic Testing for Monogenic Disorders
Preimplantation genetic testing for monogenic disorders detects specific inherited conditions. Couples who carry genetic mutations risk passing conditions to children. Cystic fibrosis, sickle cell disease, and Tay-Sachs disease represent common targets. Hundreds of conditions can be tested. The specific mutation must be known beforehand.
Before IVF begins, genetic counselors work with families. DNA samples from affected relatives help establish testing accuracy. Custom testing protocols are designed for each family’s mutations. Embryo biopsy and testing proceed similarly to aneuploidy screening. Only unaffected embryos are transferred. This virtually eliminates the risk of affected children.
Chromosomal Structural Rearrangements
Some people carry balanced chromosomal translocations. Their cells contain normal chromosome amounts but pieces are rearranged. These individuals have normal health. However, their embryos often inherit unbalanced combinations. Unbalanced embryos fail to implant or miscarry. Testing identifies embryos with balanced translocations or normal chromosomes.
Robertsonian translocations and reciprocal translocations both cause reproduction challenges. Carriers experience recurrent miscarriage and infertility. IVF with genetic testing dramatically improves their chances. Testing distinguishes balanced from unbalanced embryos. This technology has transformed outcomes for translocation carriers.
HLA Matching for Stem Cell Donation
Human leukocyte antigen matching serves a unique purpose. Families with a child needing stem cell transplant can use IVF with testing. Embryos are tested for both genetic disease status and HLA compatibility. A matched sibling’s cord blood can provide stem cells for transplant. This approach has saved lives of children with genetic blood disorders.
Ethical considerations surround this practice. Families must consider the future child’s autonomy and welfare. Most ethics boards approve when the child will be unharmed by donation. Cord blood collection at birth carries no risk to the newborn. The practice remains controversial but legally permitted in most countries.
Sex Selection and Gender Selection
Genetic testing reveals embryo sex. Sex-linked genetic conditions justify medical sex selection. Hemophilia and Duchenne muscular dystrophy affect primarily males. Selecting female embryos avoids these conditions. Non-medical sex selection for family balancing remains controversial. Many countries prohibit selection without medical indication. Individual clinics and countries have varying policies.
Is Genetic Testing Right for You?
Genetic testing decisions are complex and personal. Our genetic counselors help you understand benefits, limitations, and implications specific to your situation.
Donor Eggs, Donor Sperm, and Third-Party Reproduction
Third-party reproduction uses eggs, sperm, or embryos from donors. Some individuals cannot use their own genetic material. Donor programs help these people achieve parenthood. Egg donation assists women with poor egg quality or ovarian failure. Sperm donation helps men without sperm production or with genetic conditions. Both programs have helped thousands build families.
Egg Donation Programs
Egg donors are typically women aged 21 to 32. Extensive screening evaluates physical and psychological health. Medical history review eliminates those with hereditary conditions. Genetic testing screens for carrier status of common disorders. Psychological evaluation ensures donors understand the process and implications. Physical examination confirms reproductive health. Drug screening and infectious disease testing protect recipients.
Two egg donation models exist. Known donation involves someone the recipient knows personally. This might be a sister or friend. Anonymous donation matches recipients with unknown donors. Agencies maintain databases of screened donors. Recipients review donor profiles including photos, education, medical history, and personality information. Some programs offer identity-release donors who agree to future contact.
The egg donor undergoes ovarian stimulation and retrieval. The recipient prepares her uterus simultaneously with hormone medications. Retrieved eggs are fertilized with recipient’s partner’s sperm or donor sperm. Resulting embryos are transferred to the recipient’s uterus. Success rates with donor eggs are generally high because donors are young. Age of the recipient carrying the pregnancy matters less than egg age.
Fresh Versus Frozen Donor Egg Cycles
Fresh donor cycles synchronize donor stimulation with recipient preparation. The process requires careful coordination. Retrieved eggs are immediately fertilized and embryos transferred to the recipient. This approach offers the most eggs but requires precise timing. Cycle cancellation affects both parties if either has problems.
Frozen donor egg programs purchase previously frozen eggs. Donors undergo stimulation and retrieval. Their eggs are frozen and added to an inventory. Recipients purchase a batch of eggs when ready. Thawing occurs on demand. This model offers more flexibility and reduces coordination stress. Success rates are similar to fresh cycles with modern vitrification.
Sperm Donation Programs
Sperm donors provide samples to sperm banks. Extensive screening ensures donor health and genetic soundness. Donors undergo physical examination, genetic testing, and psychological evaluation. Infectious disease testing occurs multiple times over several months. Detailed medical and family histories are recorded. Semen analysis confirms excellent sperm parameters.
Recipients select donors from catalogs containing detailed profiles. Adult photos are typically unavailable but baby photos may be included. Educational background, occupation, interests, and personality characteristics help selection. Physical attributes including height, weight, eye color, and hair color are listed. Ethnic background matching often influences decisions. Some banks offer identity-release donors.
Donor sperm are quarantined for six months. Donors return for repeat infectious disease testing. This protocol virtually eliminates disease transmission risk. Sperm remains frozen until the quarantine period completes. Recipients purchase vials as needed. Intrauterine insemination represents the simplest use. IVF with donor sperm is used when female factors exist.
Embryo Donation
Embryo donation occurs when couples with frozen embryos offer them to others. Donors have completed their families. Rather than discarding embryos, they donate to infertile couples. Recipients undergo medical screening but need not use fertility medications. The donated frozen embryos are thawed and transferred. This offers the least expensive path to parenthood for some.
Genetic connection exists to neither parent. Some people prefer this because both partners are equal. Others find this aspect challenging. Counseling helps prospective parents explore feelings about genetic connections. Open versus anonymous embryo donation parallels egg and sperm donation models. Programs vary regarding contact and information sharing.
Legal and Ethical Considerations
Third-party reproduction involves complex legal issues. Parental rights must be clearly established before birth. Donor agreements specify no parental rights or responsibilities. Recipients become legal parents. Some jurisdictions require pre-birth legal processes. Others rely on standard adoption procedures after birth. Legal counsel specializing in reproductive law proves essential.
Disclosure to children represents an important consideration. Mental health professionals generally recommend telling children about their genetic origins. Age-appropriate discussions help children integrate this information. Many argue children have a right to know their genetic heritage. Attitudes about disclosure continue evolving as more donor-conceived adults share their perspectives.
Exploring donor options feels overwhelming. Our team guides you through every decision with compassion and expertise. Call +91-8800481100 for supportive guidance.
Gestational Surrogacy and Gestational Carrier Terminology
Surrogacy involves another woman carrying a pregnancy for intended parents. Two types exist with important distinctions. Traditional surrogacy uses the surrogate’s own egg. She is genetically related to the child. This model is rarely used today due to legal and emotional complexities. Gestational surrogacy uses the intended parents’ genetics or donor genetics. The gestational carrier has no genetic connection to the baby.
Gestational Carrier Requirements
Gestational carriers undergo rigorous screening. Age requirements typically range from 21 to 42 years. Prior successful pregnancy and delivery demonstrate reproductive capability. Current parenting shows understanding of pregnancy and childbirth. Medical evaluation confirms uterine normalcy and overall health. Psychological screening evaluates emotional stability and understanding.
Carriers must demonstrate stable life circumstances. Strong support systems help carriers through the journey. Financial stability ensures pregnancy participation isn’t economically coerced. Background checks protect intended parents. Infectious disease testing follows standard protocols. Detailed family medical history reveals genetic risks that might affect pregnancy.
The Surrogacy IVF Process
The intended mother or egg donor undergoes ovarian stimulation. Egg retrieval collects mature eggs. The intended father’s sperm or donor sperm fertilizes eggs. Embryos develop in the laboratory. Meanwhile, the gestational carrier prepares her uterus with hormone medications. Estrogen thickens the uterine lining. Progesterone prepares the lining for implantation.
When the carrier’s uterus is ready, embryo transfer occurs. The gestational carrier attends this appointment along with intended parents. One or two embryos are typically transferred. The carrier continues hormone supplementation. Pregnancy testing occurs two weeks after transfer. If pregnancy occurs, the carrier receives prenatal care. Intended parents typically attend appointments.
Types of Surrogacy Arrangements
Commercial surrogacy involves compensation beyond medical expenses. Gestational carriers receive base compensation plus additional payments. These arrangements are legal in some countries and states but prohibited in others. Altruistic surrogacy allows expense reimbursement only. No compensation for time or effort is permitted. Different jurisdictions have varying laws.
Agency-assisted surrogacy involves professional matching services. Agencies screen carriers and provide support throughout. They coordinate legal, medical, and psychological services. Independent surrogacy occurs when parties find each other directly. This reduces costs but increases coordination responsibilities. Both models work successfully with proper legal and medical support.
Surrogacy Indications
Several medical conditions necessitate gestational carriers. Uterine absence from birth or surgical removal prevents pregnancy. Severe uterine abnormalities make carrying impossible. Some medical conditions make pregnancy life-threatening. Heart disease, kidney disease, or other conditions may contraindicate pregnancy. Previous pregnancy complications might require avoiding future pregnancy.
Recurrent pregnancy loss despite normal embryos might indicate uterine factors. Repeated IVF failures suggest implantation problems. Same-sex male couples and single men need gestational carriers. These intended parents often use donor eggs as well. Surrogacy represents their path to biological parenthood.
Legal Aspects of Surrogacy
Surrogacy law varies dramatically by location. Some states have clear statutes supporting gestational surrogacy. Others have no laws or prohibit the practice. International surrogacy involves additional legal complexity. Citizenship for the baby must be carefully planned. Pre-birth orders establish parental rights in some jurisdictions. Post-birth adoption may be required elsewhere.
Surrogacy agreements detail all parties’ rights and responsibilities. Medical decision-making authority must be clear. Financial arrangements are specified. Expectations about prenatal testing and pregnancy termination require discussion. Contact during pregnancy and after birth should be addressed. Experienced reproductive attorneys protect everyone’s interests.
Considering Surrogacy?
Surrogacy involves complex medical, legal, and emotional considerations. Our experienced team guides intended parents through every step of this transformative journey.
Early Pregnancy and Pregnancy Complications Terminology
Achieving pregnancy represents only the first step toward having a baby. Many complications can occur during early pregnancy. Understanding terminology helps patients comprehend what’s happening. Clear communication with healthcare providers becomes easier with knowledge. Some pregnancy complications are minor. Others represent serious medical emergencies requiring immediate attention.
Biochemical Pregnancy
Biochemical pregnancy means positive pregnancy tests without ultrasound evidence. Blood tests detect human chorionic gonadotropin hormone. However, pregnancy fails before an ultrasound can visualize anything. This very early loss occurs before or around expected menstruation. Many women experience biochemical pregnancies without knowing. Fertility treatment increases awareness because frequent testing occurs.
Biochemical pregnancies account for about 50% of all conceptions. Most happen due to chromosomal abnormalities in the embryo. The body recognizes the problem early. No pregnancy sac develops in the uterus. Menstruation occurs normally or slightly delayed. Emotionally, these losses can be devastating despite being common. Medical treatment is rarely necessary.
Ectopic Pregnancy
Ectopic pregnancy occurs when an embryo implants outside the uterus. The fallopian tube represents the most common location. Other sites include ovary, cervix, or abdominal cavity. Ectopic pregnancies cannot develop normally. The growing embryo eventually ruptures surrounding structures. This creates a medical emergency with serious bleeding risk.
Symptoms include one-sided pelvic pain and vaginal bleeding. However, ectopic pregnancies sometimes cause no symptoms initially. Rising hCG levels without visible uterine pregnancy raises suspicion. Ultrasound confirms the diagnosis. Treatment depends on size and hCG levels. Methotrexate medication can resolve early ectopic pregnancies. Surgery becomes necessary for ruptured or large ectopic pregnancies.
Miscarriage and Pregnancy Loss
Miscarriage means pregnancy loss before 20 weeks gestation. Spontaneous abortion is the medical term but sounds harsh to patients. Early pregnancy loss occurs before 12 weeks. This accounts for most miscarriages. Late pregnancy loss happens between 12 and 20 weeks. Stillbirth refers to fetal death after 20 weeks.
Chromosomal abnormalities cause most early losses. The embryo cannot develop normally. Maternal health problems sometimes contribute. Advanced age increases miscarriage risk. Symptoms include bleeding and cramping. However, some miscarriages produce no symptoms. Ultrasound reveals absent heartbeat or development stopping. Blood hCG levels stop rising appropriately.
Several miscarriage types exist. Threatened miscarriage involves bleeding but the pregnancy continues. Inevitable miscarriage means pregnancy will definitely end. Incomplete miscarriage leaves tissue in the uterus. Complete miscarriage passes all pregnancy tissue. Missed miscarriage occurs when the embryo dies but remains in the uterus. The woman has no symptoms until tissue finally passes or intervention occurs.
Recurrent Pregnancy Loss
Recurrent pregnancy loss means two or more consecutive miscarriages. This affects about 1% of couples. Additional testing becomes appropriate. Chromosomal testing of both partners looks for translocations. Uterine anatomy evaluation identifies structural problems. Hormone testing checks thyroid and progesterone. Autoimmune testing screens for antibodies affecting pregnancy.
Antiphospholipid antibodies can cause recurrent loss. These antibodies increase blood clotting in placental vessels. Testing identifies this treatable condition. Blood thinners during pregnancy prevent future losses. Many recurrent losses have no identified cause. This proves emotionally challenging. However, most couples with unexplained recurrent loss eventually achieve successful pregnancy.
Molar Pregnancy
Molar pregnancy occurs when abnormal cells develop instead of a normal embryo. Complete molar pregnancy contains no fetal tissue. Only abnormal placental tissue grows. Partial molar pregnancy includes some fetal tissue but severe abnormalities exist. Both types cause abnormally high hCG levels. Ultrasound shows characteristic appearance of molar tissue.
Treatment involves removing molar tissue. Suction curettage evacuates the uterus. Close monitoring continues afterward because molar cells can become cancerous. Serial hCG measurements ensure levels return to zero. Pregnancy must be avoided for six months to one year. Future pregnancies carry slight increased molar pregnancy risk.
Heterotopic Pregnancy
Heterotopic pregnancy means simultaneous intrauterine and ectopic pregnancies. This is extremely rare in natural conception. Fertility treatment increases incidence because multiple embryos may implant in different locations. One embryo implants properly in the uterus. Another implants in a fallopian tube. Diagnosis can be challenging.
The ectopic portion requires treatment while preserving the uterine pregnancy. Surgical removal of the ectopic pregnancy is usually necessary. The intrauterine pregnancy may continue normally. However, risk of losing both pregnancies exists. This challenging scenario requires experienced care.
Experiencing pregnancy complications is frightening. Our compassionate team provides expert care and emotional support. Call +91-8800481100 immediately if you have concerns.
Ovulation, Menstrual Cycle, and Timing Terminology
Understanding the menstrual cycle helps explain fertility timing. The cycle begins on the first day of menstrual bleeding. Average cycles last 28 days but normal range extends from 21 to 35 days. Irregular cycles make predicting ovulation difficult. Cycle length varies primarily due to differences in the follicular phase. The luteal phase remains relatively constant at 12 to 14 days.
The Follicular Phase
The follicular phase starts with menstruation. Hormone levels are low initially. The pituitary gland increases FSH production. This stimulates several ovarian follicles to begin growing. Each follicle contains an immature egg. One follicle becomes dominant. It grows faster than others. The dominant follicle produces increasing estrogen.
Rising estrogen thickens the uterine lining. The lining prepares for potential embryo implantation. Cervical mucus becomes more abundant and stretchy. This fertile cervical mucus helps sperm travel through the cervix. As estrogen peaks, the pituitary responds with an LH surge. This surge triggers final egg maturation and ovulation.
Ovulation
Ovulation is the release of a mature egg from the ovary. The LH surge occurs 24 to 36 hours before ovulation. Home ovulation predictor kits detect this surge in urine. A positive test indicates ovulation is imminent. The mature follicle ruptures. The egg releases into the pelvic cavity. Fimbria at the fallopian tube opening capture the egg.
The egg survives only 12 to 24 hours after ovulation. Sperm must be present in the fallopian tube during this window. Sperm can survive several days in fertile cervical mucus. Having intercourse before ovulation is ideal. Sperm are waiting when the egg arrives. Some women experience mittelschmerz, or ovulation pain. This one-sided pelvic twinge occurs at ovulation time.
The Luteal Phase
After ovulation, the empty follicle transforms into the corpus luteum. This structure produces progesterone hormone. Progesterone transforms the uterine lining for implantation. The lining becomes secretory and nutrient-rich. Body temperature rises slightly due to progesterone. Basal body temperature tracking uses this temperature shift to confirm ovulation occurred.
If pregnancy occurs, the implanting embryo produces hCG. This hormone sustains the corpus luteum. Progesterone production continues. The uterine lining is maintained. Without pregnancy, the corpus luteum degenerates after about 14 days. Progesterone levels drop. The uterine lining can no longer be maintained. Menstruation begins as the lining sheds. A new cycle starts.
Ovulation Disorders
Anovulation means not ovulating. Some women rarely or never ovulate. Polycystic ovary syndrome commonly causes anovulation. Multiple small follicles develop but none become dominant. Hormone imbalances prevent ovulation. Irregular or absent periods indicate possible anovulation. Without ovulation, pregnancy cannot occur naturally.
Oligo-ovulation means infrequent ovulation. Cycles longer than 35 days suggest this problem. Hypothalamic amenorrhea occurs when stress, weight loss, or excessive exercise suppress hormones. The hypothalamus doesn’t signal the pituitary properly. Ovulation stops. Primary ovarian insufficiency means early menopause. Ovaries stop functioning before age 40. Egg supply depletes prematurely.
Ovulation Induction
Ovulation induction uses medications to trigger ovulation. Clomiphene citrate is often the first treatment. This oral medication blocks estrogen receptors. The body perceives low estrogen and increases FSH production. Follicles develop and ovulate. Letrozole works similarly but through a different mechanism. Both medications help women ovulate regularly.
Injectable gonadotropins provide stronger stimulation. These contain FSH and sometimes LH. Doctors monitor follicle development with ultrasound and blood tests. A trigger shot induces final maturation when follicles are ready. Ovulation occurs 36 to 40 hours later. Timed intercourse or insemination happens during this window. Multiple follicles may develop, increasing twin and triplet risk.
Struggling with Irregular Cycles?
Ovulation disorders are common but treatable. Our specialists identify the cause of your irregular cycles and develop personalized treatment plans to restore regular ovulation.
Intrauterine Insemination (IUI) and Artificial Insemination Terms
Intrauterine insemination is a fertility treatment placing sperm directly into the uterus. This bypasses the cervix and vagina. Sperm are deposited near the fallopian tubes. The procedure increases the number of sperm reaching eggs. IUI is simpler and less expensive than IVF. Success rates are lower but adequate for some couples.
IUI Procedure Process
IUI begins with sperm preparation. Ejaculated semen contains more than just sperm. Seminal plasma and other cells must be removed. Sperm washing separates motile sperm from debris. Only the healthiest, most motile sperm are selected. This concentrated sperm sample is used for insemination. Preparation takes one to two hours.
Meanwhile, the woman undergoes timing assessment. Natural cycle IUI occurs around spontaneous ovulation. Ovulation predictor kits help time the procedure. Stimulated IUI combines medications with insemination. Clomiphene or letrozole stimulates follicle development. Injectable medications are sometimes used. Ultrasound monitoring tracks follicle growth. A trigger shot induces final maturation.
The insemination procedure is quick and relatively painless. The woman lies in a position similar to a pap smear. A speculum allows cervical visualization. A thin catheter passes through the cervix into the uterus. Prepared sperm are slowly injected. The catheter is removed. The woman rests briefly before leaving. The entire process takes minutes. Some cramping may occur afterward.
IUI Indications and Success Factors
Several conditions make IUI appropriate. Unexplained infertility with patent tubes benefits from IUI. Mild male factor infertility responds well. Cervical mucus problems that prevent sperm passage improve with IUI. Ejaculatory dysfunction makes IUI helpful. Same-sex female couples and single women use IUI with donor sperm. Mild endometriosis patients may try IUI before advancing to IVF.
Success rates depend on multiple factors. Patient age significantly affects outcomes. Women under 35 have highest success rates. Sperm quality impacts results. At least 5 million total motile sperm are needed for adequate success. Tubal patency is essential. At least one open fallopian tube is required. Multiple IUI attempts may be necessary. Most pregnancies occur within three to four cycles.
Natural Cycle Versus Stimulated IUI
Natural cycle IUI uses no fertility medications. The woman ovulates on her own. Ovulation prediction determines timing. This approach minimizes medication side effects and costs. Success rates are somewhat lower per cycle. Natural cycles work well for younger women with regular ovulation. Donor sperm use often starts with natural cycles.
Stimulated IUI combines ovulation induction with insemination. Medications develop multiple follicles. More eggs mean higher pregnancy chances per cycle. However, multiple pregnancy risk increases significantly. Twins occur in about 10% to 20% of stimulated IUI pregnancies. Triplets and higher-order multiples are also possible. Careful monitoring limits the number of developing follicles.
IUI Versus IVF Decision-Making
IUI represents less invasive treatment than IVF. Costs are significantly lower. Physical demands are minimal. However, success rates per cycle are also lower. IUI works best for specific diagnoses. Severe male factor infertility requires IVF with ICSI. Blocked fallopian tubes necessitate IVF. Advanced maternal age reduces IUI success. Most doctors recommend IVF after three to six failed IUI cycles.
Some couples proceed directly to IVF. Severe diagnoses make IUI unlikely to succeed. When time is limited due to age, IVF maximizes efficiency. Insurance coverage sometimes influences decisions. Cost-effectiveness calculations consider cumulative success across multiple attempts. IUI may require many attempts to match one IVF cycle’s success. Individual circumstances guide treatment selection.
Wondering if IUI is right for you? Our specialists evaluate your specific situation and recommend the most effective treatment path. Call +91-8800481100 for an evaluation.
Common Fertility Diagnoses and Medical Conditions
Several medical conditions commonly impact fertility. Understanding these diagnoses helps patients comprehend their situations. Each condition has specific characteristics and treatment approaches. Some conditions affect men. Others affect women. Many have multiple treatment options available.
Polycystic Ovary Syndrome (PCOS)
Polycystic ovary syndrome is the most common hormonal disorder in reproductive-age women. The condition involves irregular ovulation or anovulation. Women with PCOS often have irregular or absent periods. Ovaries contain multiple small follicles visible on ultrasound. These follicles don’t mature and ovulate normally. Hormone imbalances include elevated androgens.
Symptoms vary widely among women. Some experience significant weight gain. Others maintain normal weight. Excess facial and body hair may develop. Acne and scalp hair thinning occur in some patients. Insulin resistance frequently accompanies PCOS. This increases type 2 diabetes risk. Not all symptoms appear in every patient.
PCOS treatment depends on whether pregnancy is desired. For conception, ovulation induction is necessary. Clomiphene citrate or letrozole help most women ovulate. Metformin improves insulin sensitivity and may help ovulation. Weight loss significantly improves symptoms and fertility. Injectable gonadotropins or IVF become necessary for some. Managing PCOS continues throughout life even after childbearing is complete.
Endometriosis
Endometriosis occurs when tissue similar to uterine lining grows outside the uterus. This tissue appears on ovaries, fallopian tubes, and pelvic organs. It responds to menstrual hormones. Bleeding occurs within these abnormal locations. Inflammation and scarring result. Pelvic pain is the hallmark symptom. Pain worsens during menstruation.
Endometriosis affects fertility through multiple mechanisms. Inflammation interferes with egg and embryo quality. Adhesions distort pelvic anatomy. Fallopian tubes may become blocked or damaged. Ovarian endometriomas are cyst-like collections of endometriosis. These may damage surrounding ovarian tissue. Even mild endometriosis affects fertility through unclear mechanisms.
Treatment depends on symptom severity and fertility goals. Pain management uses hormonal medications. Birth control pills, Depo-Provera, or GnRH agonists suppress endometriosis. However, these prevent pregnancy. Surgical excision or ablation of endometriosis lesions helps some women. Surgery can restore normal anatomy. However, endometriosis often recurs. IVF bypasses many endometriosis-related fertility problems.
Uterine Fibroids
Uterine fibroids are benign tumors in the uterine muscle. They are extremely common. Most women develop fibroids during reproductive years. Size varies from tiny to massive. Location matters more than size for fertility. Submucosal fibroids project into the uterine cavity. These significantly impair implantation. Intramural fibroids grow within the uterine wall. Large intramural fibroids may affect fertility.
Subserosal fibroids grow on the outer uterine surface. These rarely affect fertility. Many women with fibroids conceive naturally. Others experience recurrent miscarriage or implantation failure. Treatment depends on symptoms and fertility impact. Expectant management works when fibroids cause no problems. Myomectomy surgically removes fibroids while preserving the uterus.
Multiple surgical approaches exist for myomectomy. Hysteroscopic myomectomy removes submucosal fibroids through the cervix. Laparoscopic or robotic myomectomy uses small abdominal incisions. Abdominal myomectomy requires a larger incision. Surgery carries some risks. Fibroids may recur. In severe cases, hysterectomy becomes necessary. Uterine artery embolization shrinks fibroids but may impact fertility.
Diminished Ovarian Reserve
Diminished ovarian reserve means fewer eggs remain in the ovaries. Age is the primary cause. Egg quantity declines throughout life. Quality also deteriorates with age. Some women experience premature decline. Genetic factors, surgeries, or treatments may damage ovaries. Testing reveals low AMH and elevated FSH levels. Antral follicle count is reduced on ultrasound.
Response to ovarian stimulation is poor. Fewer eggs develop despite high medication doses. Egg quality is often reduced. Fewer embryos develop. Pregnancy rates per cycle are lower. Treatment options are limited. Higher medication doses may help some patients. However, stimulation cannot restore the original egg supply. Many women with diminished reserve require donor eggs eventually.
Male Factor Infertility Causes
Varicocele is enlarged veins in the scrotum. Blood pools in these veins. Testicular temperature rises. Sperm production decreases. Varicocele is the most common correctable male infertility cause. Physical examination detects larger varicoceles. Ultrasound confirms diagnosis. Surgical repair improves sperm parameters in many men.
Hormonal imbalances affect sperm production. Low testosterone or elevated prolactin require evaluation. Thyroid disorders influence fertility. Genetic conditions including Klinefelter syndrome cause severe male infertility. Y chromosome microdeletions impair sperm production. Cystic fibrosis gene mutations cause congenital absence of vas deferens. Previous infections or injuries damage reproductive structures. Treatment depends on the specific cause identified.
Recently Diagnosed with a Fertility Condition?
Understanding your diagnosis is the first step toward effective treatment. Our specialists explain your condition thoroughly and create personalized treatment plans to optimize your fertility.
Reproductive Anatomy and System Terminology
Understanding reproductive anatomy helps explain fertility processes. The female reproductive system has multiple components working together. Each structure serves specific functions. Problems with any component can impair fertility. The male reproductive system is equally complex. Proper function requires coordination of multiple organs and processes.
Female Reproductive Organs
The uterus is a muscular organ where pregnancy develops. The endometrium is the uterine lining. This tissue thickens monthly in preparation for implantation. Without pregnancy, the endometrium sheds during menstruation. The myometrium is the thick muscular uterine wall. These muscles expand during pregnancy and contract during labor. The cervix forms the lower uterine opening. It connects the uterus to the vagina.
The vagina is a muscular canal connecting external and internal reproductive structures. The vulva includes external female genital structures. The ovaries are paired organs containing eggs. They produce estrogen and progesterone hormones. Follicles are fluid-filled sacs within ovaries. Each follicle contains one egg. Thousands of follicles exist at birth. This number continuously decreases.
Fallopian tubes connect ovaries to the uterus. Eggs travel through these tubes after ovulation. Fertilization typically occurs in the fallopian tube. The tube’s interior has hair-like projections called cilia. These cilia move the egg toward the uterus. The fimbria are finger-like projections at the tube opening. They capture released eggs. Any damage to tubes impairs fertility significantly.
Male Reproductive Organs
The testes are paired organs producing sperm and testosterone. Seminiferous tubules within testes are where sperm production occurs. This process takes approximately 70 days from start to finish. Sertoli cells support developing sperm. Leydig cells produce testosterone hormone. Both cell types are essential for normal function.
The epididymis is a coiled tube attached to each testis. Sperm mature and gain motility here. Sperm storage also occurs in the epididymis. The vas deferens is a muscular tube transporting sperm. It connects the epididymis to the urethra. Vasectomy cuts or blocks the vas deferens. This prevents sperm from entering ejaculate.
The prostate gland produces fluid contributing to semen. This fluid nourishes and protects sperm. Seminal vesicles produce additional seminal fluid. The bulbourethral glands also contribute secretions. Semen is the complete ejaculated fluid. It contains sperm plus fluids from these glands. The urethra carries semen through the penis during ejaculation. It also carries urine from the bladder.
Hormonal Control of Reproduction
The hypothalamus is a brain region controlling reproduction. It releases gonadotropin-releasing hormone in pulses. This signals the pituitary gland. The pituitary is a small gland at the brain’s base. It produces follicle-stimulating hormone and luteinizing hormone. These hormones travel through blood to reproductive organs. They control egg and sperm production.
Negative feedback regulates hormone levels. Rising sex hormones signal the brain to reduce gonadotropin production. Decreasing sex hormones trigger increased gonadotropin release. This system maintains hormone balance. Disruptions cause fertility problems. Medications can manipulate this system for fertility treatment.
Gametes and Fertilization
Gametes are reproductive cells. Eggs and sperm are gametes. Normal cells contain 46 chromosomes in 23 pairs. Gametes undergo meiosis during development. This special cell division halves the chromosome number. Mature gametes contain 23 single chromosomes. Fertilization combines egg and sperm chromosomes. The resulting embryo has 46 chromosomes again.
Sex chromosomes determine biological sex. Eggs always carry an X chromosome. Sperm carry either X or Y chromosomes. XX combinations produce females. XY combinations produce males. Chromosome abnormalities occur when meiosis errors happen. Extra or missing chromosomes usually prevent normal development. This causes most early pregnancy losses.
Questions about your reproductive anatomy or how specific structures work? Our team explains anatomy in understandable terms. Call +91-8800481100 for detailed explanations.
Fertility Medications and Treatment Drug Terminology
Fertility treatment involves various medications. Understanding drug names and purposes reduces confusion. Most drugs have both generic and brand names. Generic names describe the actual medication. Brand names are specific to manufacturers. Medications fall into several categories based on function and purpose.
Ovulation Induction Medications
Clomiphene citrate is an oral medication stimulating ovulation. Brand names include Clomid and Serophene. The drug blocks estrogen receptors in the brain. The body responds by increasing FSH production. This stimulates follicle development. Typical doses range from 50 to 150 milligrams daily. Treatment lasts five days starting early in the cycle.
Letrozole is an aromatase inhibitor used off-label for fertility. Brand name Femara treats breast cancer at higher doses. For fertility, 2.5 to 5 milligrams daily for five days is standard. Letrozole blocks estrogen production. The pituitary increases FSH similarly to clomiphene. Some evidence suggests letrozole works better than clomiphene for PCOS patients. Side effects are generally minimal.
Injectable Gonadotropins
Injectable FSH medications stimulate multiple follicle development. Follitropin alfa and follitropin beta are recombinant forms. Brand names include Gonal-F and Follistim. These identical medications are produced through genetic engineering. Urofollitropin is purified from postmenopausal women’s urine. Bravelle is one brand name. Human menopausal gonadotropin contains both FSH and LH. Menopur is a common brand.
Dosing varies based on patient age, weight, and ovarian reserve. Starting doses range from 75 to 300 international units daily. Doctors adjust doses based on monitoring results. Injections continue for 8 to 14 days typically. Medications come as powders requiring mixing. Prefilled pens simplify administration. Patients self-administer subcutaneous injections. Proper injection technique training is essential.
Trigger Shot Medications
Human chorionic gonadotropin triggers final egg maturation. This hormone mimics the natural LH surge. Brand names include Pregnyl, Novarel, and Ovidrel. Ovidrel is recombinant hCG requiring no mixing. Others use urinary hCG requiring reconstitution. Dosing ranges from 5,000 to 10,000 international units. Timing must be precise. Egg retrieval or insemination occurs 35 to 36 hours later.
GnRH agonist can also trigger ovulation. Lupron is a commonly used brand. Triggering with agonist reduces OHSS risk significantly. However, luteal phase support becomes more critical. Some protocols use dual trigger. Both hCG and agonist are given together. This may improve egg maturity while reducing complications.
GnRH Antagonists and Agonists
Gonadotropin-releasing hormone antagonists prevent premature ovulation. Ganirelix and cetrotide are commonly used. These medications block GnRH receptors immediately. Daily injections start when follicles reach a certain size. Treatment continues until trigger shot administration. Antagonists don’t require suppression before stimulation begins. This shortens treatment time.
GnRH agonists serve multiple purposes. Initially, they stimulate hormone production. After several days, they suppress the pituitary gland. This suppression prevents premature ovulation. Lupron is the most common agonist. Long protocols use agonist suppression before stimulation. Short protocols add agonist during stimulation. The same medication works differently depending on timing and dosing.
Progesterone Supplementation
Progesterone support is essential after egg retrieval. Natural production is inadequate because retrieval removes progesterone-producing cells. Multiple progesterone formulations exist. Intramuscular progesterone in oil is very effective but requires daily injections. The injection is painful and causes muscle soreness. Subcutaneous progesterone formulations reduce discomfort.
Vaginal progesterone includes suppositories, gels, and tablets. Crinone, Endometrin, and generic forms are available. These avoid injections but can be messy. Oral progesterone is less commonly used. It requires higher doses because of liver metabolism. Combination approaches use multiple formulations together. Progesterone continues through early pregnancy if conception occurs.
Supportive Medications
Estrogen patches or pills prepare the uterine lining. These are essential for frozen embryo transfer cycles. Baby aspirin reduces blood clotting slightly. Some doctors prescribe this to improve implantation. Antibiotics prevent infection after egg retrieval. Doxycycline is commonly prescribed. Prenatal vitamins should begin before conception. Folic acid prevents neural tube defects. Additional supplements may be recommended based on individual needs.
Overwhelmed by Medication Instructions?
We understand fertility medications can feel overwhelming. Our nursing staff provides detailed medication teaching and remains available throughout your treatment cycle to answer questions.
Laboratory Values, Results, and Testing Terminology
Laboratory testing provides crucial fertility information. Understanding test results helps patients participate in treatment decisions. Numbers alone don’t tell the complete story. Context matters. Age affects interpretation of many values. Lab ranges represent averages. Individual results require personalized assessment. Doctors consider multiple factors when interpreting results.
Hormone Level Interpretation
FSH measured on cycle day two or three assesses ovarian reserve. Normal values vary by laboratory. Generally, FSH under 10 IU/L is considered normal. Values between 10 and 15 suggest diminishing reserve. FSH exceeding 15 indicates significant reserve decline. Very high FSH over 20 means poor reserve. Treatment response will likely be minimal. However, FSH fluctuates between cycles. One elevated value doesn’t define prognosis definitively.
Anti-Müllerian hormone provides additional reserve information. This value remains stable throughout the menstrual cycle. Higher AMH indicates more remaining follicles. AMH over 2.0 nanograms per milliliter suggests good reserve. Values between 1.0 and 2.0 are adequate. AMH under 1.0 indicates diminished reserve. Very low AMH below 0.5 suggests poor stimulation response. Extremely high AMH may indicate PCOS.
Estradiol on day two or three should be low. Elevated baseline estradiol suggests a cyst or premature follicle development. This may affect cycle timing. During stimulation, rising estradiol confirms follicle growth. Each mature follicle produces approximately 200 to 300 picograms per milliliter. Very high estradiol increases OHSS risk. Doctors may adjust medication or delay trigger if estradiol rises too quickly.
Progesterone Testing
Progesterone confirms ovulation occurred. Testing happens approximately one week after suspected ovulation. This coincides with mid-luteal phase. Progesterone over 3 nanograms per milliliter confirms ovulation. Values over 10 indicate strong ovulation. Very low progesterone suggests anovulation or poor corpus luteum function. During IVF cycles, progesterone supplementation makes this test unhelpful. Exogenous progesterone raises levels regardless of natural production.
Pregnancy Testing
Quantitative hCG measures exact hormone levels. This blood test is more sensitive than home pregnancy tests. Values over 5 mIU/mL indicate pregnancy. In early pregnancy, hCG should double every 48 to 72 hours. Slow rises suggest problems. Very high hCG may indicate multiple pregnancies or molar pregnancy. Serial measurements track pregnancy progression. Declining hCG indicates pregnancy loss.
Qualitative pregnancy tests give yes or no results. Home pregnancy tests work this way. Blood qualitative tests are rarely necessary. Most clinics use quantitative testing. The first hCG test occurs about two weeks after embryo transfer. This is called beta day. A second test follows 48 hours later. The doubling pattern predicts viability better than single values.
Semen Analysis Parameters
Volume measures ejaculated semen quantity. Normal range is 1.5 to 5 milliliters. Low volume suggests collection problems or anatomical issues. Very high volume dilutes sperm concentration. Concentration counts sperm per milliliter. Normal concentration exceeds 15 million per milliliter. Total count multiplies concentration by volume. At least 39 million total sperm should be present.
Motility percentages indicate moving sperm. Progressive motility specifically counts forward-moving sperm. At least 32% should show progressive movement. Total motility includes all movement types. Morphology assesses sperm shape. By strict Kruger criteria, at least 4% should have normal form. Some labs use WHO criteria where 15% normal is acceptable. Additional tests evaluate vitality, DNA fragmentation, and antibodies when indicated.
Ultrasound Measurements
Antral follicle count is performed during baseline ultrasound. Small follicles measuring 2 to 10 millimeters are counted on both ovaries. Total count predicts ovarian reserve and stimulation response. Count over 12 suggests good reserve. Count between 6 and 12 is adequate. Counts under 6 indicate poor reserve. Very high counts over 25 may indicate PCOS.
During stimulation, growing follicles are measured. Mature follicles typically measure 16 to 22 millimeters. Lead follicles are the largest ones. When multiple follicles reach maturity, trigger timing is determined. Endometrial thickness is also measured. Adequate thickness is at least 7 millimeters. Thicker linings over 8 to 10 millimeters are better. Pattern also matters. Trilaminar appearance is ideal.
Confused about your test results? Our specialists interpret your numbers and explain what they mean for your fertility treatment. Call +91-8800481100 to discuss your results.
Understanding Fertility Treatment Success Rates and Statistics
Success rates help patients understand treatment effectiveness. However, statistics can be misleading without context. Multiple ways exist to calculate and report success. Understanding these differences prevents unrealistic expectations. Individual results vary significantly from published averages. Personal factors affect outcomes more than general statistics suggest.
Pregnancy Rate Versus Live Birth Rate
Pregnancy rate measures positive pregnancy tests. This includes all pregnancies regardless of outcome. Some pregnancies end in miscarriage. Chemical pregnancies count toward pregnancy rates. Clinical pregnancy rate requires ultrasound confirmation of gestational sac. This provides more meaningful information than biochemical pregnancy rates alone.
Live birth rate represents actual babies born. This is the most important statistic for patients. Live birth rate per cycle started counts all attempts. This includes cycles canceled before egg retrieval. Live birth rate per embryo transfer excludes canceled cycles. This inflates apparent success. Cumulative live birth rate across multiple cycles provides realistic expectations. Most live births occur within three to four IVF attempts.
Age and Success Rates
Maternal age dramatically affects all fertility treatment outcomes. Women under 35 have highest success rates. Pregnancy and live birth rates decrease gradually from 35 to 40. Decline accelerates after 40. By age 44, success rates drop below 5% per cycle in most cases. Donor eggs eliminate age effects on success rates. The egg donor’s age matters while recipient age matters much less.
Male age also influences outcomes though less dramatically. Advanced paternal age increases miscarriage risk. Sperm DNA damage accumulates with age. However, men maintain fertility potential much longer than women. Combined parental age matters when both partners are older. Success rates reflect the more significant limiting factor.
Singleton Versus Multiple Birth Rates
Multiple pregnancy rates vary with embryo number transferred. Single embryo transfer dramatically reduces twin risk. Some embryos split naturally creating identical twins. This occurs in about 1% to 2% of IVF pregnancies. Transferring two embryos increases twin chances significantly. Both embryos may implant. One might split creating triplets from two embryos.
Multiple pregnancies carry higher risks. Preterm birth is much more common. Low birth weight affects most multiples. Pregnancy complications increase. Mother’s health risks rise substantially. Neonatal intensive care is often necessary. Long-term developmental issues are more frequent. These risks explain the push toward single embryo transfer.
Miscarriage Rates
Miscarriage risk increases with maternal age. Women under 35 have approximately 15% miscarriage risk. This rises to 25% by age 40. After 42, miscarriage exceeds 50% of pregnancies. Chromosome abnormalities cause most age-related losses. Genetic testing of embryos may reduce miscarriage rates. However, testing doesn’t eliminate all loss. Other factors contribute beyond chromosomes.
Clinic-Specific Versus National Statistics
Individual clinics report their own success rates. These vary considerably between programs. Patient population differences partly explain variations. Clinics accepting harder cases may show lower success rates. However, they might actually provide better care. Cherry-picking easy cases inflates statistics artificially. National databases compile all clinic data. This provides better overall picture of expected success.
The Society for Assisted Reproductive Technology maintains U.S. data. Centers for Disease Control also publishes fertility clinic success rates. These reports lag two years behind current cycles. Treatment protocols evolve continuously. Historical data may not reflect current practices. Individual consultation provides more accurate personal predictions than published statistics.
Want to Know Your Personal Success Probability?
General statistics don’t tell your individual story. We assess your specific situation and provide personalized success predictions based on your unique circumstances.
Moving Forward: Your Next Steps in Understanding Fertility
Understanding infertility terminology empowers you throughout your fertility journey. Medical language no longer needs to feel intimidating. You now have knowledge to communicate effectively with your healthcare team. This comprehensive glossary covered hundreds of terms across all aspects of fertility care. From basic anatomy to complex procedures, you’ve gained essential vocabulary.
Knowledge transforms anxiety into confidence. When doctors discuss your treatment plan, you’ll understand the reasoning. Test results make sense in context. Medication purposes become clear. Procedure steps are no longer mysterious. This understanding helps you participate actively in decisions affecting your reproductive future.
Remember that every fertility journey is unique. Your specific situation requires personalized evaluation and treatment. While this glossary provides general information, individual circumstances vary widely. What works for one person may not work for another. Age, diagnosis, and personal factors all influence appropriate treatment approaches.
describes couples who have never achieved pregnancy despite trying. These individuals have no history of conception. Secondary infertility occurs when couples have conceived before but cannot achieve another pregnancy. Both types present unique emotional and medical challenges requiring different approaches to treatment and support.
Male Factor Infertility
Male infertility accounts for about 40% of all infertility cases. Issues with sperm production, sperm quality, or sperm delivery create challenges for conception. Sperm count refers to the number of sperm cells present in a semen sample. Low sperm count means fewer than 15 million sperm per milliliter of semen. Sperm motility describes how well sperm move and swim toward an egg.
Motile sperm are those capable of forward movement necessary to reach and fertilize an egg. Poor sperm motility reduces the chances of natural conception. Sperm morphology examines the shape and structure of sperm cells. Abnormally shaped sperm may struggle to penetrate an egg successfully. These three factors combine to determine overall male fertility potential.
Female Factor Infertility
Female infertility involves issues with the reproductive tract, hormonal imbalances, or egg quality problems. The fallopian tubes transport eggs from ovaries to the uterus. Blocked or damaged fallopian tubes prevent sperm from reaching eggs. Ovulation disorders represent another common cause. When ovaries don’t release eggs regularly, conception becomes difficult or impossible.
The uterus must provide a healthy environment for embryo implantation. Structural problems or uterine lining issues interfere with this process. Age significantly impacts female fertility through declining egg quantity and quality. These factors often work in combination rather than isolation.
Confused about whether your situation involves male or female factors? Call +91-8800481100 to discuss your specific circumstances with our fertility experts.
Assisted Reproductive Technology (ART): Comprehensive Overview
Assisted reproductive technology encompasses all fertility treatments where both eggs and sperm are handled outside the body. These procedures help couples overcome various infertility obstacles. ART represents modern medicine’s most advanced approach to treating reproductive challenges. Success rates vary based on age, diagnosis, and specific procedure type.
What ART Includes
The assisted reproductive technology category includes several distinct procedures. In vitro fertilization forms the foundation of most ART treatments. Intracytoplasmic sperm injection represents a specialized technique within IVF. Egg and embryo cryopreservation allow patients to preserve fertility for future use. Donor egg and donor sperm programs help patients who cannot use their own genetic material.
Gestational surrogacy involves another woman carrying a pregnancy. Preimplantation genetic testing screens embryos for genetic conditions. Each ART procedure addresses specific fertility challenges. Some patients require multiple procedures combined to achieve pregnancy.
ART Success Factors
Multiple factors influence ART outcomes. Patient age plays the most significant role in success rates. Women under 35 typically experience higher success rates than older patients. The cause of infertility affects treatment effectiveness. Previous pregnancy history provides insight into potential outcomes. Embryo quality directly impacts implantation chances.
The number of embryos transferred influences both success rates and multiple pregnancy risks. Clinic experience and laboratory quality matter enormously. Proper patient selection and protocol customization improve results. Lifestyle factors including weight, smoking, and alcohol consumption affect outcomes.
Understanding Live Birth Rates
Live birth rates represent the ultimate measure of ART success. This statistic counts actual babies born, not just positive pregnancy tests. One cycle of treatment may or may not result in a live birth. Multiple cycles often prove necessary to achieve the desired outcome. National registries track live birth statistics to help patients understand realistic expectations.
Age-specific live birth rates provide the most relevant information for individual patients. Cumulative live birth rates across multiple cycles offer a more complete picture. Understanding these statistics helps patients set realistic expectations and make informed decisions about treatment continuation.
Ready to Explore ART Options?
Our team specializes in personalized assisted reproductive technology treatment plans. We’ll help you understand which procedures offer the best chance of success for your unique situation.
In Vitro Fertilization (IVF) Terminology Explained
In vitro fertilization literally means fertilization in glass. This process occurs in a laboratory dish rather than inside a woman’s body. IVF has helped millions of couples worldwide achieve pregnancy since the first successful procedure in 1978. The technique involves multiple steps that span several weeks from start to finish.
The IVF Cycle Process
An IVF cycle begins with ovarian stimulation. Women take hormone medications to encourage multiple eggs to develop simultaneously. Normally, ovaries release one egg per month. Stimulation increases this number significantly. More eggs provide more opportunities for successful fertilization and embryo development.
Monitoring occurs throughout stimulation through ultrasound examinations and blood tests. These procedures track follicle growth and hormone levels. When eggs reach maturity, a trigger shot initiates final egg maturation. Egg retrieval takes place approximately 36 hours after the trigger injection. This minor surgical procedure collects eggs from the ovaries using ultrasound guidance.
Fertilization and Embryo Development
After retrieval, embryologists combine eggs with sperm in the laboratory. Conventional IVF places sperm and eggs together, allowing natural fertilization to occur. Intracytoplasmic sperm injection involves directly injecting a single sperm into each egg. This technique helps when sperm quality or quantity issues exist.
Fertilized eggs become embryos. Embryologists observe embryo development over several days. Day 3 embryos typically contain six to eight cells. Blastocyst embryos develop by day 5 or 6, containing over 100 cells. Many clinics prefer transferring blastocysts because they better predict successful implantation.
Embryo Transfer and Implantation
Embryo transfer places one or more embryos into the uterus. This simple procedure requires no anesthesia and resembles a pap smear. A thin catheter carries embryos through the cervix into the uterine cavity. Ultrasound guidance ensures proper placement. The entire process takes just minutes to complete.
Implantation occurs when an embryo attaches to the uterine lining. This natural process happens several days after transfer. Not all transferred embryos successfully implant. The two-week wait between transfer and pregnancy testing creates significant emotional stress for patients. Blood tests measure hormone levels to confirm pregnancy.
Luteal Phase Support
The luteal phase occurs after ovulation or egg retrieval. Progesterone hormone prepares the uterine lining for implantation. IVF patients typically require progesterone supplementation because retrieval removes cells that normally produce this hormone. Support continues through early pregnancy if conception occurs. Various progesterone formulations include injections, vaginal suppositories, and oral medications.
Want to understand how IVF could work for you specifically? Our specialists explain every step in detail. Call +91-8800481100 for a personalized consultation.
Intracytoplasmic Sperm Injection and Specialized Fertility Procedures
Intracytoplasmic sperm injection revolutionized treatment for severe male infertility. This precise procedure involves injecting a single sperm directly into an egg’s cytoplasm. ICSI bypasses many natural barriers that prevent fertilization. The technique requires highly skilled embryologists and specialized equipment. Success rates often match or exceed conventional IVF fertilization methods.
When ICSI Is Recommended
Several conditions indicate ICSI as the preferred fertilization method. Very low sperm count makes conventional fertilization unlikely to succeed. Poor sperm motility prevents sperm from reaching eggs naturally. Abnormal sperm morphology reduces penetration capability. Previous IVF cycles with failed fertilization suggest ICSI for subsequent attempts.
Using frozen sperm often requires ICSI because freezing may damage some sperm cells. Surgical sperm extraction from the testes always necessitates ICSI. Eggs with thick outer shells may benefit from direct injection. Some clinics recommend ICSI for all patients to maximize fertilization rates.
The ICSI Procedure Step by Step
ICSI begins with careful sperm selection. Embryologists choose the healthiest-appearing sperm under high magnification. Individual sperm selection considers morphology and motility. The selected sperm is drawn into a tiny glass needle. Simultaneously, an egg is held steady with a holding pipette.
The injection needle pierces the egg membrane carefully. The sperm is deposited directly into the egg’s interior. This process repeats for each mature egg. After injection, eggs are cultured in specialized media. Fertilization confirmation occurs the following day when two pronuclei appear inside the egg.
Advanced Sperm Selection Techniques
Several advanced techniques help identify the best sperm for ICSI. Physiological intracytoplasmic sperm injection uses hyaluronic acid to select mature sperm. This substance mimics the egg’s natural coating. Only mature sperm bind to hyaluronic acid, improving selection accuracy. The technique may reduce miscarriage rates and increase pregnancy success.
Intracellular sperm morphology assessment examines sperm at extremely high magnification. This allows embryologists to detect subtle structural abnormalities invisible under standard magnification. Selecting sperm with normal internal structures may improve outcomes. Research continues regarding the effectiveness of these enhanced selection methods.
Surgical Sperm Retrieval
Some men produce sperm but cannot ejaculate them naturally. Surgical procedures extract sperm directly from the reproductive tract. Testicular sperm extraction removes tissue samples from the testes. Percutaneous epididymal sperm aspiration uses a needle to withdraw sperm from the epididymis. Microsurgical epididymal sperm aspiration uses surgical magnification for precise sperm collection.
These procedures help men with obstructive azoospermia achieve biological fatherhood. Non-obstructive azoospermia occurs when sperm production is severely reduced. Microdissection testicular sperm extraction can sometimes find small pockets of sperm production. All surgically retrieved sperm require ICSI for fertilization.
Is ICSI Right for Your Situation?
Male factor infertility doesn’t mean you can’t have biological children. Our specialists evaluate your specific case and recommend the most effective fertilization approach.
Understanding Eggs, Embryos, and Early Development
The oocyte is the medical term for an egg cell. Women are born with approximately one to two million oocytes. This number decreases throughout life. By puberty, about 300,000 remain. Each month during reproductive years, one oocyte typically matures and releases during ovulation. Only about 300 to 400 eggs ever ovulate during a woman’s lifetime.
Egg Maturity and Quality
Not all retrieved eggs are mature enough for fertilization. Germinal vesicle eggs are immature and cannot be fertilized immediately. Metaphase I eggs have started maturation but haven’t completed the process. Metaphase II eggs are fully mature and ready for fertilization. Only metaphase II eggs can be used for IVF or ICSI procedures.
Egg quality significantly impacts fertilization success and embryo development. Age affects egg quality more than any other factor. Chromosomal abnormalities increase in eggs as women age. These abnormalities reduce fertilization rates, increase miscarriage risk, and lower live birth chances. No current treatment can reverse age-related egg quality decline.
Fertilization and Early Embryo Development
Fertilization occurs when sperm penetrates the egg. The fertilized egg is now called a zygote. Cell division begins within hours of fertilization. The two-cell embryo appears approximately 24 hours after fertilization. Division continues rapidly over subsequent days. Four-cell embryos develop around day two. Eight-cell embryos form by day three.
At this stage, embryos are also called cleavage-stage embryos. Each cell is called a blastomere. The embryo continues dividing while traveling toward the uterus in natural conception. In IVF, embryos remain in culture dishes during this time. Embryologists monitor development daily, documenting progress and quality.
Morula and Blastocyst Formation
Around day four, the embryo becomes a morula. This solid ball of cells contains 16 to 32 blastomeres. The cells begin to differentiate and organize. A cavity forms inside the embryo. When this cavity appears, the embryo becomes a blastocyst. This typically occurs on day five or six after fertilization.
Blastocysts contain two distinct cell types. The inner cell mass will become the fetus. The trophectoderm cells will form the placenta and membranes. Blastocyst quality grading assesses expansion, inner cell mass, and trophectoderm. Higher-grade blastocysts have better implantation potential. Many clinics prefer transferring blastocysts rather than earlier-stage embryos.
Embryo Grading Systems
Embryologists use standardized grading systems to evaluate embryo quality. Day-three embryos are graded based on cell number, cell symmetry, and fragmentation percentage. Ideal day-three embryos have seven to nine cells with minimal fragmentation. Even cell sizes indicate better developmental potential.
Blastocyst grading considers three factors. Expansion describes how developed the cavity is. Inner cell mass quality predicts fetal development potential. Trophectoderm quality indicates placental development capability. A blastocyst might be graded as 4AA, with the number representing expansion and letters representing inner cell mass and trophectoderm quality respectively.
Questions about egg or embryo quality in your specific case? Our embryologists can explain your results in understandable terms. Call +91-8800481100 today.
Fertility Hormones and Ovarian Stimulation Protocols
Hormones control the entire reproductive process. Understanding these chemical messengers helps explain fertility treatments. Follicle-stimulating hormone prompts follicles in the ovaries to grow. Each follicle contains one egg. FSH levels naturally rise at the beginning of each menstrual cycle. Injectable FSH medications stimulate multiple follicles simultaneously during IVF.
Key Reproductive Hormones
Luteinizing hormone triggers ovulation in natural cycles. The LH surge signals an egg to complete maturation and release. IVF uses injectable human chorionic gonadotropin as a trigger shot because it mimics LH. This hormone also supports early pregnancy. Pregnancy tests detect hCG in urine or blood.
Estrogen rises as follicles grow. This hormone thickens the uterine lining and prepares it for implantation. High estrogen levels during stimulation indicate good follicle development. Progesterone prepares and maintains the uterine lining after ovulation. It prevents menstruation if pregnancy occurs. All IVF patients need progesterone supplementation after egg retrieval.
Anti-Müllerian hormone helps predict ovarian reserve. Higher AMH levels suggest more remaining eggs. This hormone helps doctors determine appropriate medication dosing. Thyroid hormones affect overall reproductive function. Thyroid disorders require correction before starting fertility treatment.
Ovarian Stimulation Medications
Multiple protocols exist for ovarian stimulation. Gonadotropins are injectable hormones that stimulate follicle development. These medications contain FSH alone or FSH combined with LH. Daily injections continue for approximately 8 to 14 days. Doctors adjust doses based on ultrasound and blood test results.
GnRH antagonists prevent premature ovulation during stimulation. These daily injections typically start when follicles reach a certain size. GnRH agonists can serve similar functions but work differently. Long protocols use agonists to suppress natural hormones before stimulation begins. Short protocols start agonists during stimulation.
Oral medications sometimes supplement injection protocols. Clomiphene citrate stimulates natural FSH production. Letrozole also promotes follicle development through a different mechanism. These drugs cost less than injectables but generally produce fewer eggs. Some mild stimulation protocols combine oral medications with low-dose injectables.
Monitoring During Stimulation
Patients undergo frequent monitoring throughout stimulation. Transvaginal ultrasound examinations measure follicle size and count. Technicians measure each visible follicle on the ovaries. Mature follicles typically measure 16 to 22 millimeters in diameter. Blood tests measure estrogen levels to confirm follicle maturity.
Rising estrogen indicates growing follicles. Very high estrogen levels may indicate ovarian hyperstimulation risk. Monitoring continues every few days initially, then daily as follicles mature. This information guides medication adjustments and determines trigger shot timing. Precise timing ensures optimal egg maturity at retrieval.
Trigger Shot and Final Maturation
The trigger shot initiates final egg maturation. This injection must occur at precisely the right time. Too early means eggs won’t be mature enough. Too late risks ovulation before retrieval. Most triggers use human chorionic gonadotropin. Some protocols use a GnRH agonist as a trigger instead.
Egg retrieval is scheduled exactly 35 to 36 hours after the trigger injection. This timing allows eggs to complete maturation without releasing from follicles. Patients must follow trigger timing instructions precisely. Even a few hours difference can significantly impact results. Some protocols use a dual trigger combining both hCG and GnRH agonist.
Concerned About Medication Protocols?
Every stimulation protocol is customized to your unique situation. Our specialists explain medication purposes, side effects, and what to expect throughout treatment.
Diagnostic Procedures and Fertility Testing Terminology
Accurate diagnosis forms the foundation of effective fertility treatment. Multiple tests evaluate both male and female fertility factors. Results guide treatment selection and predict success probabilities. Most fertility workups begin with simple, non-invasive tests before progressing to more complex procedures.
Semen Analysis Basics
Semen analysis provides essential information about male fertility potential. This test evaluates several key parameters. Volume measures the amount of ejaculated semen. Normal volume ranges from 1.5 to 5 milliliters. Low volume may indicate collection problems or reproductive tract issues. Very high volume can dilute sperm concentration.
Sperm concentration counts sperm cells per milliliter. Normal concentration exceeds 15 million per milliliter. Total sperm count multiplies concentration by volume. At least 39 million total sperm should be present. Oligospermia describes low sperm count. Azoospermia means no sperm are present in the ejaculate.
Progressive motility measures forward-moving sperm. At least 32% of sperm should show progressive movement. Total motility includes all moving sperm. Normal morphology examines sperm shape. At least 4% should have normal form by strict criteria. Vitality testing determines the percentage of live sperm. Additional tests may examine DNA fragmentation or antibody presence.
Female Hormone Testing
Baseline hormone testing occurs early in the menstrual cycle. Day two or three blood tests measure FSH, LH, and estradiol. These values help assess ovarian reserve and function. Elevated FSH suggests diminished ovarian reserve. High LH relative to FSH may indicate polycystic ovary syndrome. Low estradiol combined with high FSH confirms poor ovarian function.
Anti-Müllerian hormone testing can occur any cycle day. This value remains relatively stable throughout the cycle. Higher AMH suggests better ovarian reserve. Very low AMH indicates few remaining eggs. Very high AMH may indicate PCOS. Progesterone testing confirms ovulation occurrence. This test happens approximately one week after suspected ovulation.
Thyroid function tests evaluate TSH and sometimes additional thyroid hormones. Abnormal thyroid function interferes with conception and pregnancy maintenance. Prolactin elevation can prevent ovulation. This hormone requires evaluation if menstrual irregularity exists. Additional hormone tests may be ordered based on symptoms and initial results.
Ultrasound Examinations
Transvaginal ultrasound provides detailed views of reproductive organs. This procedure uses a probe inserted into the vagina. Sound waves create images of the uterus and ovaries. Antral follicle count occurs during early cycle baseline ultrasounds. Small follicles visible on the ovaries predict how many eggs might develop during stimulation.
Ultrasound evaluates uterine structure and identifies abnormalities. Fibroids appear as solid masses in or around the uterus. Polyps project into the uterine cavity. Both conditions may interfere with implantation. Ultrasound measures uterine lining thickness. Adequate thickness is necessary for embryo implantation. Ovarian cysts or masses are also visible on ultrasound.
Hysterosalpingography and Tubal Assessment
Hysterosalpingography evaluates fallopian tube patency. This X-ray procedure involves injecting contrast dye through the cervix. The dye fills the uterine cavity and flows through fallopian tubes. X-rays capture images as dye progresses. Open tubes allow dye to spill into the abdominal cavity. Blocked tubes prevent dye passage.
This test causes some discomfort but typically requires no anesthesia. Cramping occurs as dye distends the uterus. The procedure also reveals uterine shape abnormalities. Saline infusion sonography serves as an alternative tube-testing method. Ultrasound visualization occurs as sterile saline fills the uterus. Bubbles visible in the tubes suggest patency.
Advanced Diagnostic Procedures
Hysteroscopy allows direct visualization of the uterine cavity. A thin telescope passes through the cervix. Doctors can diagnose and sometimes treat abnormalities during the same procedure. Small polyps or fibroids can be removed. Uterine septum or scar tissue can be corrected. Hysteroscopy requires anesthesia and occurs in a surgical setting.
Laparoscopy examines the pelvic cavity. Small incisions allow camera insertion. Surgeons can evaluate endometriosis, adhesions, and tubal damage. Treatment often occurs simultaneously with diagnosis. This procedure requires general anesthesia and brief recovery time. Endometrial biopsy samples uterine lining tissue. Analysis can reveal inflammation, infection, or timing issues.
Not sure which tests you need? Our fertility specialists create comprehensive diagnostic plans tailored to your situation. Call +91-8800481100 to schedule your evaluation.
Embryo Transfer Procedures and Implantation Process
Embryo transfer represents the final step in an IVF cycle. This relatively simple procedure determines whether treatment succeeds or fails. Despite its simplicity, technique matters significantly. Proper placement increases implantation chances. The procedure causes minimal discomfort and requires no anesthesia in most cases.
Transfer Day Preparation
Patients arrive with a moderately full bladder. Fluid in the bladder helps visualize the uterus via abdominal ultrasound. Too much fluid causes discomfort. Too little prevents adequate visualization. Timing the perfect bladder fullness requires practice. Some patients need to partially empty their bladder before the procedure.
The embryologist prepares selected embryos for transfer. Embryos are loaded into a thin, flexible catheter. This occurs in the laboratory moments before transfer. The physician confirms embryo presence in the catheter. Mock transfers may have occurred previously to plan the exact approach. Cervical preparation with gentle cleaning occurs before catheter insertion.
The Transfer Procedure
The patient lies in a position similar to a pap smear. A speculum allows cervical visualization. The physician gently passes the catheter through the cervix into the uterus. Abdominal ultrasound guidance ensures correct placement. The ideal location is in the upper-middle portion of the uterine cavity. The catheter should not touch the fundus or upper uterine wall.
When proper position is confirmed, embryos are gently expelled. The catheter remains still during expulsion to avoid dislodging embryos. After transfer, the catheter is carefully removed. The embryologist immediately examines the catheter under a microscope. This confirms all embryos left the catheter successfully. Occasionally, an embryo adheres to the catheter and requires a second transfer attempt.
Fresh Versus Frozen Embryo Transfer
Fresh embryo transfer occurs three to six days after egg retrieval. The patient’s body is still recovering from stimulation. Hormone levels remain elevated from trigger medications. Some evidence suggests frozen embryo transfer may achieve higher success rates. The uterus may be more receptive when not recently stimulated.
Frozen embryo transfer occurs in a subsequent cycle. All embryos undergo cryopreservation after development. The uterus is prepared using hormone medications or timed with natural ovulation. This approach allows time for recovery from stimulation. Genetic testing can be completed before transfer. Remaining embryos are available for future attempts without repeating stimulation and retrieval.
Single Versus Multiple Embryo Transfer
Single embryo transfer means placing one embryo in the uterus. This approach minimizes multiple pregnancy risk. Twin and higher-order pregnancies carry increased complications. Mother and babies face higher risks than singleton pregnancies. Many clinics now advocate for elective single embryo transfer, especially for younger patients with high-quality embryos.
Multiple embryo transfer increases pregnancy chances per cycle. However, it also dramatically increases multiple pregnancy risk. Two embryos might both implant. One embryo might split into identical twins. The result could be triplets from transferring two embryos. Pregnancy and delivery complications rise significantly with multiples. Individual circumstances guide the decision about embryo number.
Understanding Implantation
Implantation begins several days after transfer. The blastocyst hatches from its protective shell. Trophectoderm cells begin invading the uterine lining. This process triggers local immune and vascular changes. Blood vessels form connections between the embryo and maternal circulation. The developing placenta secretes human chorionic gonadotropin hormone.
Successful implantation depends on embryo quality and uterine receptivity. Both must be optimal at the same time. The implantation window refers to the brief period when the uterus is receptive. This typically occurs six to ten days after ovulation. Timing embryo transfer to coincide with this window maximizes success. Endometrial receptivity testing can identify individual window timing.
Questions About Your Upcoming Transfer?
Embryo transfer is a crucial moment in your fertility journey. Our team ensures you understand every aspect of the procedure and what to expect during the waiting period afterward.
Cryopreservation and Fertility Preservation Options
Cryopreservation freezes biological materials for future use. Fertility preservation allows people to maintain reproductive options. Cancer patients often preserve fertility before treatment begins. People delaying parenthood can freeze eggs or embryos. Same-sex couples and single individuals utilize preservation as part of family planning. The technology has improved dramatically over recent decades.
Egg Freezing (Oocyte Cryopreservation)
Egg freezing preserves unfertilized eggs. Women undergo ovarian stimulation identical to IVF. Egg retrieval collects mature eggs. Embryologists immediately freeze eggs using vitrification. This ultra-rapid freezing prevents ice crystal formation. Ice crystals would damage delicate egg structures. Vitrification transformed egg freezing from experimental to routine.
Frozen eggs can remain in storage for many years. No definitive time limit exists for storage. Eggs thawed years later show similar survival and fertilization rates. Success depends heavily on age at freezing. Younger eggs yield better outcomes than eggs frozen at older ages. Women typically freeze 15 to 20 eggs for optimal pregnancy chances later.
When ready to use frozen eggs, they undergo thawing. Survival rates exceed 90% with vitrification. Surviving eggs are fertilized using ICSI. This injection technique prevents damage from sperm binding attempts. Embryos develop normally and are transferred or refrozen. Pregnancy rates from frozen eggs approach those from fresh eggs when age-matched.
Embryo Freezing (Embryo Cryopreservation)
Embryo cryopreservation freezes fertilized embryos. Couples often create more embryos than can be safely transferred. Excess embryos are frozen for future use. This avoids repeating stimulation and egg retrieval for subsequent pregnancies. Embryo freezing has been performed successfully since the 1980s. Modern vitrification techniques yield excellent survival rates.
Embryos can be frozen at various developmental stages. Day-three embryos and blastocysts both freeze well. Many clinics prefer freezing blastocysts because they tolerate the process well. Survival rates often exceed 95%. Frozen embryo transfer cycles require endometrial preparation. The uterine lining must be ready to receive the embryo.
Patients taking ownership of frozen embryos face important decisions. Embryos can be stored indefinitely while paying storage fees. They can be used for future pregnancies. Donation to other couples helps those unable to create embryos. Donation to research contributes to scientific advancement. Some choose to discontinue storage and allow thawing without use.
Sperm Freezing (Sperm Cryopreservation)
Sperm freezing preserves male fertility. Men facing cancer treatment routinely freeze sperm beforehand. Chemotherapy and radiation often damage sperm production. Recovery may take years or never occur. Frozen sperm provides insurance against this risk. Men with declining sperm parameters may bank sperm while quality remains adequate.
The freezing process involves mixing sperm with cryoprotectant. Samples are divided into multiple vials. This allows using one vial per IVF attempt. Slow-rate freezing or vitrification methods both work for sperm. Sperm tolerate freezing better than eggs. Survival rates typically remain high. Frozen sperm can be used for IUI, IVF, or ICSI procedures.
Surgically retrieved sperm are always frozen initially. Recovery typically yields limited quantities. Using fresh surgical sperm would require perfect IVF cycle timing. Freezing allows flexible scheduling. Men undergoing vasectomy might freeze sperm beforehand. This maintains options if life circumstances change later.
Ovarian Tissue Freezing
Ovarian tissue cryopreservation represents the newest preservation option. This technique helps women who cannot delay cancer treatment for ovarian stimulation. Surgery removes all or part of one ovary. The outer cortex containing thousands of immature follicles is frozen. After cancer recovery, tissue can be transplanted back. This has resulted in natural pregnancies and live births.
The procedure remains experimental but shows promise. Very young girls can preserve fertility before puberty. Mature eggs cannot be retrieved before puberty begins. Ovarian tissue offers their only current preservation option. Future technologies may allow maturing frozen follicles in the laboratory. Research continues to improve outcomes with this innovative approach.
Considering fertility preservation? Time matters, especially before medical treatments. Call +91-8800481100 to discuss your options and timing.
Preimplantation Genetic Testing and Screening
Preimplantation genetic testing examines embryos before transfer. This technology identifies chromosomal abnormalities and genetic conditions. Testing helps select the healthiest embryos. Several types of genetic testing serve different purposes. All require embryo biopsy to obtain cells for analysis. Results guide embryo selection decisions.
Preimplantation Genetic Testing for Aneuploidies
Preimplantation genetic testing for aneuploidies screens chromosome numbers. Humans normally have 23 pairs of chromosomes. Aneuploid embryos have extra or missing chromosomes. Most aneuploidies prevent implantation or cause early miscarriage. Down syndrome results from an extra chromosome 21. Monosomy X causes Turner syndrome. Trisomy 13 and 18 are usually fatal.
Aneuploidy increases dramatically with maternal age. Women under 35 produce about 50% aneuploid embryos. This percentage exceeds 75% by age 40. Testing identifies chromosomally normal embryos for transfer. This may reduce miscarriage rates and improve live birth rates per transfer. However, testing doesn’t improve overall cumulative success rates across multiple cycles.
The biopsy typically occurs at the blastocyst stage. Several trophectoderm cells are removed. These will become placenta, not baby. The embryo is frozen while awaiting results. Testing usually takes one to two weeks. Normal embryos are transferred in a subsequent cycle. Mosaic embryos contain both normal and abnormal cells. Management of mosaic results requires careful counseling.
Preimplantation Genetic Testing for Monogenic Disorders
Preimplantation genetic testing for monogenic disorders detects specific inherited conditions. Couples who carry genetic mutations risk passing conditions to children. Cystic fibrosis, sickle cell disease, and Tay-Sachs disease represent common targets. Hundreds of conditions can be tested. The specific mutation must be known beforehand.
Before IVF begins, genetic counselors work with families. DNA samples from affected relatives help establish testing accuracy. Custom testing protocols are designed for each family’s mutations. Embryo biopsy and testing proceed similarly to aneuploidy screening. Only unaffected embryos are transferred. This virtually eliminates the risk of affected children.
Chromosomal Structural Rearrangements
Some people carry balanced chromosomal translocations. Their cells contain normal chromosome amounts but pieces are rearranged. These individuals have normal health. However, their embryos often inherit unbalanced combinations. Unbalanced embryos fail to implant or miscarry. Testing identifies embryos with balanced translocations or normal chromosomes.
Robertsonian translocations and reciprocal translocations both cause reproduction challenges. Carriers experience recurrent miscarriage and infertility. IVF with genetic testing dramatically improves their chances. Testing distinguishes balanced from unbalanced embryos. This technology has transformed outcomes for translocation carriers.
HLA Matching for Stem Cell Donation
Human leukocyte antigen matching serves a unique purpose. Families with a child needing stem cell transplant can use IVF with testing. Embryos are tested for both genetic disease status and HLA compatibility. A matched sibling’s cord blood can provide stem cells for transplant. This approach has saved lives of children with genetic blood disorders.
Ethical considerations surround this practice. Families must consider the future child’s autonomy and welfare. Most ethics boards approve when the child will be unharmed by donation. Cord blood collection at birth carries no risk to the newborn. The practice remains controversial but legally permitted in most countries.
Sex Selection and Gender Selection
Genetic testing reveals embryo sex. Sex-linked genetic conditions justify medical sex selection. Hemophilia and Duchenne muscular dystrophy affect primarily males. Selecting female embryos avoids these conditions. Non-medical sex selection for family balancing remains controversial. Many countries prohibit selection without medical indication. Individual clinics and countries have varying policies.
Is Genetic Testing Right for You?
Genetic testing decisions are complex and personal. Our genetic counselors help you understand benefits, limitations, and implications specific to your situation.
Donor Eggs, Donor Sperm, and Third-Party Reproduction
Third-party reproduction uses eggs, sperm, or embryos from donors. Some individuals cannot use their own genetic material. Donor programs help these people achieve parenthood. Egg donation assists women with poor egg quality or ovarian failure. Sperm donation helps men without sperm production or with genetic conditions. Both programs have helped thousands build families.
Egg Donation Programs
Egg donors are typically women aged 21 to 32. Extensive screening evaluates physical and psychological health. Medical history review eliminates those with hereditary conditions. Genetic testing screens for carrier status of common disorders. Psychological evaluation ensures donors understand the process and implications. Physical examination confirms reproductive health. Drug screening and infectious disease testing protect recipients.
Two egg donation models exist. Known donation involves someone the recipient knows personally. This might be a sister or friend. Anonymous donation matches recipients with unknown donors. Agencies maintain databases of screened donors. Recipients review donor profiles including photos, education, medical history, and personality information. Some programs offer identity-release donors who agree to future contact.
The egg donor undergoes ovarian stimulation and retrieval. The recipient prepares her uterus simultaneously with hormone medications. Retrieved eggs are fertilized with recipient’s partner’s sperm or donor sperm. Resulting embryos are transferred to the recipient’s uterus. Success rates with donor eggs are generally high because donors are young. Age of the recipient carrying the pregnancy matters less than egg age.
Fresh Versus Frozen Donor Egg Cycles
Fresh donor cycles synchronize donor stimulation with recipient preparation. The process requires careful coordination. Retrieved eggs are immediately fertilized and embryos transferred to the recipient. This approach offers the most eggs but requires precise timing. Cycle cancellation affects both parties if either has problems.
Frozen donor egg programs purchase previously frozen eggs. Donors undergo stimulation and retrieval. Their eggs are frozen and added to an inventory. Recipients purchase a batch of eggs when ready. Thawing occurs on demand. This model offers more flexibility and reduces coordination stress. Success rates are similar to fresh cycles with modern vitrification.
Sperm Donation Programs
Sperm donors provide samples to sperm banks. Extensive screening ensures donor health and genetic soundness. Donors undergo physical examination, genetic testing, and psychological evaluation. Infectious disease testing occurs multiple times over several months. Detailed medical and family histories are recorded. Semen analysis confirms excellent sperm parameters.
Recipients select donors from catalogs containing detailed profiles. Adult photos are typically unavailable but baby photos may be included. Educational background, occupation, interests, and personality characteristics help selection. Physical attributes including height, weight, eye color, and hair color are listed. Ethnic background matching often influences decisions. Some banks offer identity-release donors.
Donor sperm are quarantined for six months. Donors return for repeat infectious disease testing. This protocol virtually eliminates disease transmission risk. Sperm remains frozen until the quarantine period completes. Recipients purchase vials as needed. Intrauterine insemination represents the simplest use. IVF with donor sperm is used when female factors exist.
Embryo Donation
Embryo donation occurs when couples with frozen embryos offer them to others. Donors have completed their families. Rather than discarding embryos, they donate to infertile couples. Recipients undergo medical screening but need not use fertility medications. The donated frozen embryos are thawed and transferred. This offers the least expensive path to parenthood for some.
Genetic connection exists to neither parent. Some people prefer this because both partners are equal. Others find this aspect challenging. Counseling helps prospective parents explore feelings about genetic connections. Open versus anonymous embryo donation parallels egg and sperm donation models. Programs vary regarding contact and information sharing.
Legal and Ethical Considerations
Third-party reproduction involves complex legal issues. Parental rights must be clearly established before birth. Donor agreements specify no parental rights or responsibilities. Recipients become legal parents. Some jurisdictions require pre-birth legal processes. Others rely on standard adoption procedures after birth. Legal counsel specializing in reproductive law proves essential.
Disclosure to children represents an important consideration. Mental health professionals generally recommend telling children about their genetic origins. Age-appropriate discussions help children integrate this information. Many argue children have a right to know their genetic heritage. Attitudes about disclosure continue evolving as more donor-conceived adults share their perspectives.
Exploring donor options feels overwhelming. Our team guides you through every decision with compassion and expertise. Call +91-8800481100 for supportive guidance.
Gestational Surrogacy and Gestational Carrier Terminology
Surrogacy involves another woman carrying a pregnancy for intended parents. Two types exist with important distinctions. Traditional surrogacy uses the surrogate’s own egg. She is genetically related to the child. This model is rarely used today due to legal and emotional complexities. Gestational surrogacy uses the intended parents’ genetics or donor genetics. The gestational carrier has no genetic connection to the baby.
Gestational Carrier Requirements
Gestational carriers undergo rigorous screening. Age requirements typically range from 21 to 42 years. Prior successful pregnancy and delivery demonstrate reproductive capability. Current parenting shows understanding of pregnancy and childbirth. Medical evaluation confirms uterine normalcy and overall health. Psychological screening evaluates emotional stability and understanding.
Carriers must demonstrate stable life circumstances. Strong support systems help carriers through the journey. Financial stability ensures pregnancy participation isn’t economically coerced. Background checks protect intended parents. Infectious disease testing follows standard protocols. Detailed family medical history reveals genetic risks that might affect pregnancy.
The Surrogacy IVF Process
The intended mother or egg donor undergoes ovarian stimulation. Egg retrieval collects mature eggs. The intended father’s sperm or donor sperm fertilizes eggs. Embryos develop in the laboratory. Meanwhile, the gestational carrier prepares her uterus with hormone medications. Estrogen thickens the uterine lining. Progesterone prepares the lining for implantation.
When the carrier’s uterus is ready, embryo transfer occurs. The gestational carrier attends this appointment along with intended parents. One or two embryos are typically transferred. The carrier continues hormone supplementation. Pregnancy testing occurs two weeks after transfer. If pregnancy occurs, the carrier receives prenatal care. Intended parents typically attend appointments.
Types of Surrogacy Arrangements
Commercial surrogacy involves compensation beyond medical expenses. Gestational carriers receive base compensation plus additional payments. These arrangements are legal in some countries and states but prohibited in others. Altruistic surrogacy allows expense reimbursement only. No compensation for time or effort is permitted. Different jurisdictions have varying laws.
Agency-assisted surrogacy involves professional matching services. Agencies screen carriers and provide support throughout. They coordinate legal, medical, and psychological services. Independent surrogacy occurs when parties find each other directly. This reduces costs but increases coordination responsibilities. Both models work successfully with proper legal and medical support.
Surrogacy Indications
Several medical conditions necessitate gestational carriers. Uterine absence from birth or surgical removal prevents pregnancy. Severe uterine abnormalities make carrying impossible. Some medical conditions make pregnancy life-threatening. Heart disease, kidney disease, or other conditions may contraindicate pregnancy. Previous pregnancy complications might require avoiding future pregnancy.
Recurrent pregnancy loss despite normal embryos might indicate uterine factors. Repeated IVF failures suggest implantation problems. Same-sex male couples and single men need gestational carriers. These intended parents often use donor eggs as well. Surrogacy represents their path to biological parenthood.
Legal Aspects of Surrogacy
Surrogacy law varies dramatically by location. Some states have clear statutes supporting gestational surrogacy. Others have no laws or prohibit the practice. International surrogacy involves additional legal complexity. Citizenship for the baby must be carefully planned. Pre-birth orders establish parental rights in some jurisdictions. Post-birth adoption may be required elsewhere.
Surrogacy agreements detail all parties’ rights and responsibilities. Medical decision-making authority must be clear. Financial arrangements are specified. Expectations about prenatal testing and pregnancy termination require discussion. Contact during pregnancy and after birth should be addressed. Experienced reproductive attorneys protect everyone’s interests.
Considering Surrogacy?
Surrogacy involves complex medical, legal, and emotional considerations. Our experienced team guides intended parents through every step of this transformative journey.
Early Pregnancy and Pregnancy Complications Terminology
Achieving pregnancy represents only the first step toward having a baby. Many complications can occur during early pregnancy. Understanding terminology helps patients comprehend what’s happening. Clear communication with healthcare providers becomes easier with knowledge. Some pregnancy complications are minor. Others represent serious medical emergencies requiring immediate attention.
Biochemical Pregnancy
Biochemical pregnancy means positive pregnancy tests without ultrasound evidence. Blood tests detect human chorionic gonadotropin hormone. However, pregnancy fails before an ultrasound can visualize anything. This very early loss occurs before or around expected menstruation. Many women experience biochemical pregnancies without knowing. Fertility treatment increases awareness because frequent testing occurs.
Biochemical pregnancies account for about 50% of all conceptions. Most happen due to chromosomal abnormalities in the embryo. The body recognizes the problem early. No pregnancy sac develops in the uterus. Menstruation occurs normally or slightly delayed. Emotionally, these losses can be devastating despite being common. Medical treatment is rarely necessary.
Ectopic Pregnancy
Ectopic pregnancy occurs when an embryo implants outside the uterus. The fallopian tube represents the most common location. Other sites include ovary, cervix, or abdominal cavity. Ectopic pregnancies cannot develop normally. The growing embryo eventually ruptures surrounding structures. This creates a medical emergency with serious bleeding risk.
Symptoms include one-sided pelvic pain and vaginal bleeding. However, ectopic pregnancies sometimes cause no symptoms initially. Rising hCG levels without visible uterine pregnancy raises suspicion. Ultrasound confirms the diagnosis. Treatment depends on size and hCG levels. Methotrexate medication can resolve early ectopic pregnancies. Surgery becomes necessary for ruptured or large ectopic pregnancies.
Miscarriage and Pregnancy Loss
Miscarriage means pregnancy loss before 20 weeks gestation. Spontaneous abortion is the medical term but sounds harsh to patients. Early pregnancy loss occurs before 12 weeks. This accounts for most miscarriages. Late pregnancy loss happens between 12 and 20 weeks. Stillbirth refers to fetal death after 20 weeks.
Chromosomal abnormalities cause most early losses. The embryo cannot develop normally. Maternal health problems sometimes contribute. Advanced age increases miscarriage risk. Symptoms include bleeding and cramping. However, some miscarriages produce no symptoms. Ultrasound reveals absent heartbeat or development stopping. Blood hCG levels stop rising appropriately.
Several miscarriage types exist. Threatened miscarriage involves bleeding but the pregnancy continues. Inevitable miscarriage means pregnancy will definitely end. Incomplete miscarriage leaves tissue in the uterus. Complete miscarriage passes all pregnancy tissue. Missed miscarriage occurs when the embryo dies but remains in the uterus. The woman has no symptoms until tissue finally passes or intervention occurs.
Recurrent Pregnancy Loss
Recurrent pregnancy loss means two or more consecutive miscarriages. This affects about 1% of couples. Additional testing becomes appropriate. Chromosomal testing of both partners looks for translocations. Uterine anatomy evaluation identifies structural problems. Hormone testing checks thyroid and progesterone. Autoimmune testing screens for antibodies affecting pregnancy.
Antiphospholipid antibodies can cause recurrent loss. These antibodies increase blood clotting in placental vessels. Testing identifies this treatable condition. Blood thinners during pregnancy prevent future losses. Many recurrent losses have no identified cause. This proves emotionally challenging. However, most couples with unexplained recurrent loss eventually achieve successful pregnancy.
Molar Pregnancy
Molar pregnancy occurs when abnormal cells develop instead of a normal embryo. Complete molar pregnancy contains no fetal tissue. Only abnormal placental tissue grows. Partial molar pregnancy includes some fetal tissue but severe abnormalities exist. Both types cause abnormally high hCG levels. Ultrasound shows characteristic appearance of molar tissue.
Treatment involves removing molar tissue. Suction curettage evacuates the uterus. Close monitoring continues afterward because molar cells can become cancerous. Serial hCG measurements ensure levels return to zero. Pregnancy must be avoided for six months to one year. Future pregnancies carry slight increased molar pregnancy risk.
Heterotopic Pregnancy
Heterotopic pregnancy means simultaneous intrauterine and ectopic pregnancies. This is extremely rare in natural conception. Fertility treatment increases incidence because multiple embryos may implant in different locations. One embryo implants properly in the uterus. Another implants in a fallopian tube. Diagnosis can be challenging.
The ectopic portion requires treatment while preserving the uterine pregnancy. Surgical removal of the ectopic pregnancy is usually necessary. The intrauterine pregnancy may continue normally. However, risk of losing both pregnancies exists. This challenging scenario requires experienced care.
Experiencing pregnancy complications is frightening. Our compassionate team provides expert care and emotional support. Call +91-8800481100 immediately if you have concerns.
Ovulation, Menstrual Cycle, and Timing Terminology
Understanding the menstrual cycle helps explain fertility timing. The cycle begins on the first day of menstrual bleeding. Average cycles last 28 days but normal range extends from 21 to 35 days. Irregular cycles make predicting ovulation difficult. Cycle length varies primarily due to differences in the follicular phase. The luteal phase remains relatively constant at 12 to 14 days.
The Follicular Phase
The follicular phase starts with menstruation. Hormone levels are low initially. The pituitary gland increases FSH production. This stimulates several ovarian follicles to begin growing. Each follicle contains an immature egg. One follicle becomes dominant. It grows faster than others. The dominant follicle produces increasing estrogen.
Rising estrogen thickens the uterine lining. The lining prepares for potential embryo implantation. Cervical mucus becomes more abundant and stretchy. This fertile cervical mucus helps sperm travel through the cervix. As estrogen peaks, the pituitary responds with an LH surge. This surge triggers final egg maturation and ovulation.
Ovulation
Ovulation is the release of a mature egg from the ovary. The LH surge occurs 24 to 36 hours before ovulation. Home ovulation predictor kits detect this surge in urine. A positive test indicates ovulation is imminent. The mature follicle ruptures. The egg releases into the pelvic cavity. Fimbria at the fallopian tube opening capture the egg.
The egg survives only 12 to 24 hours after ovulation. Sperm must be present in the fallopian tube during this window. Sperm can survive several days in fertile cervical mucus. Having intercourse before ovulation is ideal. Sperm are waiting when the egg arrives. Some women experience mittelschmerz, or ovulation pain. This one-sided pelvic twinge occurs at ovulation time.
The Luteal Phase
After ovulation, the empty follicle transforms into the corpus luteum. This structure produces progesterone hormone. Progesterone transforms the uterine lining for implantation. The lining becomes secretory and nutrient-rich. Body temperature rises slightly due to progesterone. Basal body temperature tracking uses this temperature shift to confirm ovulation occurred.
If pregnancy occurs, the implanting embryo produces hCG. This hormone sustains the corpus luteum. Progesterone production continues. The uterine lining is maintained. Without pregnancy, the corpus luteum degenerates after about 14 days. Progesterone levels drop. The uterine lining can no longer be maintained. Menstruation begins as the lining sheds. A new cycle starts.
Ovulation Disorders
Anovulation means not ovulating. Some women rarely or never ovulate. Polycystic ovary syndrome commonly causes anovulation. Multiple small follicles develop but none become dominant. Hormone imbalances prevent ovulation. Irregular or absent periods indicate possible anovulation. Without ovulation, pregnancy cannot occur naturally.
Oligo-ovulation means infrequent ovulation. Cycles longer than 35 days suggest this problem. Hypothalamic amenorrhea occurs when stress, weight loss, or excessive exercise suppress hormones. The hypothalamus doesn’t signal the pituitary properly. Ovulation stops. Primary ovarian insufficiency means early menopause. Ovaries stop functioning before age 40. Egg supply depletes prematurely.
Ovulation Induction
Ovulation induction uses medications to trigger ovulation. Clomiphene citrate is often the first treatment. This oral medication blocks estrogen receptors. The body perceives low estrogen and increases FSH production. Follicles develop and ovulate. Letrozole works similarly but through a different mechanism. Both medications help women ovulate regularly.
Injectable gonadotropins provide stronger stimulation. These contain FSH and sometimes LH. Doctors monitor follicle development with ultrasound and blood tests. A trigger shot induces final maturation when follicles are ready. Ovulation occurs 36 to 40 hours later. Timed intercourse or insemination happens during this window. Multiple follicles may develop, increasing twin and triplet risk.
Struggling with Irregular Cycles?
Ovulation disorders are common but treatable. Our specialists identify the cause of your irregular cycles and develop personalized treatment plans to restore regular ovulation.
Intrauterine Insemination (IUI) and Artificial Insemination Terms
Intrauterine insemination is a fertility treatment placing sperm directly into the uterus. This bypasses the cervix and vagina. Sperm are deposited near the fallopian tubes. The procedure increases the number of sperm reaching eggs. IUI is simpler and less expensive than IVF. Success rates are lower but adequate for some couples.
IUI Procedure Process
IUI begins with sperm preparation. Ejaculated semen contains more than just sperm. Seminal plasma and other cells must be removed. Sperm washing separates motile sperm from debris. Only the healthiest, most motile sperm are selected. This concentrated sperm sample is used for insemination. Preparation takes one to two hours.
Meanwhile, the woman undergoes timing assessment. Natural cycle IUI occurs around spontaneous ovulation. Ovulation predictor kits help time the procedure. Stimulated IUI combines medications with insemination. Clomiphene or letrozole stimulates follicle development. Injectable medications are sometimes used. Ultrasound monitoring tracks follicle growth. A trigger shot induces final maturation.
The insemination procedure is quick and relatively painless. The woman lies in a position similar to a pap smear. A speculum allows cervical visualization. A thin catheter passes through the cervix into the uterus. Prepared sperm are slowly injected. The catheter is removed. The woman rests briefly before leaving. The entire process takes minutes. Some cramping may occur afterward.
IUI Indications and Success Factors
Several conditions make IUI appropriate. Unexplained infertility with patent tubes benefits from IUI. Mild male factor infertility responds well. Cervical mucus problems that prevent sperm passage improve with IUI. Ejaculatory dysfunction makes IUI helpful. Same-sex female couples and single women use IUI with donor sperm. Mild endometriosis patients may try IUI before advancing to IVF.
Success rates depend on multiple factors. Patient age significantly affects outcomes. Women under 35 have highest success rates. Sperm quality impacts results. At least 5 million total motile sperm are needed for adequate success. Tubal patency is essential. At least one open fallopian tube is required. Multiple IUI attempts may be necessary. Most pregnancies occur within three to four cycles.
Natural Cycle Versus Stimulated IUI
Natural cycle IUI uses no fertility medications. The woman ovulates on her own. Ovulation prediction determines timing. This approach minimizes medication side effects and costs. Success rates are somewhat lower per cycle. Natural cycles work well for younger women with regular ovulation. Donor sperm use often starts with natural cycles.
Stimulated IUI combines ovulation induction with insemination. Medications develop multiple follicles. More eggs mean higher pregnancy chances per cycle. However, multiple pregnancy risk increases significantly. Twins occur in about 10% to 20% of stimulated IUI pregnancies. Triplets and higher-order multiples are also possible. Careful monitoring limits the number of developing follicles.
IUI Versus IVF Decision-Making
IUI represents less invasive treatment than IVF. Costs are significantly lower. Physical demands are minimal. However, success rates per cycle are also lower. IUI works best for specific diagnoses. Severe male factor infertility requires IVF with ICSI. Blocked fallopian tubes necessitate IVF. Advanced maternal age reduces IUI success. Most doctors recommend IVF after three to six failed IUI cycles.
Some couples proceed directly to IVF. Severe diagnoses make IUI unlikely to succeed. When time is limited due to age, IVF maximizes efficiency. Insurance coverage sometimes influences decisions. Cost-effectiveness calculations consider cumulative success across multiple attempts. IUI may require many attempts to match one IVF cycle’s success. Individual circumstances guide treatment selection.
Wondering if IUI is right for you? Our specialists evaluate your specific situation and recommend the most effective treatment path. Call +91-8800481100 for an evaluation.
Common Fertility Diagnoses and Medical Conditions
Several medical conditions commonly impact fertility. Understanding these diagnoses helps patients comprehend their situations. Each condition has specific characteristics and treatment approaches. Some conditions affect men. Others affect women. Many have multiple treatment options available.
Polycystic Ovary Syndrome (PCOS)
Polycystic ovary syndrome is the most common hormonal disorder in reproductive-age women. The condition involves irregular ovulation or anovulation. Women with PCOS often have irregular or absent periods. Ovaries contain multiple small follicles visible on ultrasound. These follicles don’t mature and ovulate normally. Hormone imbalances include elevated androgens.
Symptoms vary widely among women. Some experience significant weight gain. Others maintain normal weight. Excess facial and body hair may develop. Acne and scalp hair thinning occur in some patients. Insulin resistance frequently accompanies PCOS. This increases type 2 diabetes risk. Not all symptoms appear in every patient.
PCOS treatment depends on whether pregnancy is desired. For conception, ovulation induction is necessary. Clomiphene citrate or letrozole help most women ovulate. Metformin improves insulin sensitivity and may help ovulation. Weight loss significantly improves symptoms and fertility. Injectable gonadotropins or IVF become necessary for some. Managing PCOS continues throughout life even after childbearing is complete.
Endometriosis
Endometriosis occurs when tissue similar to uterine lining grows outside the uterus. This tissue appears on ovaries, fallopian tubes, and pelvic organs. It responds to menstrual hormones. Bleeding occurs within these abnormal locations. Inflammation and scarring result. Pelvic pain is the hallmark symptom. Pain worsens during menstruation.
Endometriosis affects fertility through multiple mechanisms. Inflammation interferes with egg and embryo quality. Adhesions distort pelvic anatomy. Fallopian tubes may become blocked or damaged. Ovarian endometriomas are cyst-like collections of endometriosis. These may damage surrounding ovarian tissue. Even mild endometriosis affects fertility through unclear mechanisms.
Treatment depends on symptom severity and fertility goals. Pain management uses hormonal medications. Birth control pills, Depo-Provera, or GnRH agonists suppress endometriosis. However, these prevent pregnancy. Surgical excision or ablation of endometriosis lesions helps some women. Surgery can restore normal anatomy. However, endometriosis often recurs. IVF bypasses many endometriosis-related fertility problems.
Uterine Fibroids
Uterine fibroids are benign tumors in the uterine muscle. They are extremely common. Most women develop fibroids during reproductive years. Size varies from tiny to massive. Location matters more than size for fertility. Submucosal fibroids project into the uterine cavity. These significantly impair implantation. Intramural fibroids grow within the uterine wall. Large intramural fibroids may affect fertility.
Subserosal fibroids grow on the outer uterine surface. These rarely affect fertility. Many women with fibroids conceive naturally. Others experience recurrent miscarriage or implantation failure. Treatment depends on symptoms and fertility impact. Expectant management works when fibroids cause no problems. Myomectomy surgically removes fibroids while preserving the uterus.
Multiple surgical approaches exist for myomectomy. Hysteroscopic myomectomy removes submucosal fibroids through the cervix. Laparoscopic or robotic myomectomy uses small abdominal incisions. Abdominal myomectomy requires a larger incision. Surgery carries some risks. Fibroids may recur. In severe cases, hysterectomy becomes necessary. Uterine artery embolization shrinks fibroids but may impact fertility.
Diminished Ovarian Reserve
Diminished ovarian reserve means fewer eggs remain in the ovaries. Age is the primary cause. Egg quantity declines throughout life. Quality also deteriorates with age. Some women experience premature decline. Genetic factors, surgeries, or treatments may damage ovaries. Testing reveals low AMH and elevated FSH levels. Antral follicle count is reduced on ultrasound.
Response to ovarian stimulation is poor. Fewer eggs develop despite high medication doses. Egg quality is often reduced. Fewer embryos develop. Pregnancy rates per cycle are lower. Treatment options are limited. Higher medication doses may help some patients. However, stimulation cannot restore the original egg supply. Many women with diminished reserve require donor eggs eventually.
Male Factor Infertility Causes
Varicocele is enlarged veins in the scrotum. Blood pools in these veins. Testicular temperature rises. Sperm production decreases. Varicocele is the most common correctable male infertility cause. Physical examination detects larger varicoceles. Ultrasound confirms diagnosis. Surgical repair improves sperm parameters in many men.
Hormonal imbalances affect sperm production. Low testosterone or elevated prolactin require evaluation. Thyroid disorders influence fertility. Genetic conditions including Klinefelter syndrome cause severe male infertility. Y chromosome microdeletions impair sperm production. Cystic fibrosis gene mutations cause congenital absence of vas deferens. Previous infections or injuries damage reproductive structures. Treatment depends on the specific cause identified.
Recently Diagnosed with a Fertility Condition?
Understanding your diagnosis is the first step toward effective treatment. Our specialists explain your condition thoroughly and create personalized treatment plans to optimize your fertility.
Reproductive Anatomy and System Terminology
Understanding reproductive anatomy helps explain fertility processes. The female reproductive system has multiple components working together. Each structure serves specific functions. Problems with any component can impair fertility. The male reproductive system is equally complex. Proper function requires coordination of multiple organs and processes.
Female Reproductive Organs
The uterus is a muscular organ where pregnancy develops. The endometrium is the uterine lining. This tissue thickens monthly in preparation for implantation. Without pregnancy, the endometrium sheds during menstruation. The myometrium is the thick muscular uterine wall. These muscles expand during pregnancy and contract during labor. The cervix forms the lower uterine opening. It connects the uterus to the vagina.
The vagina is a muscular canal connecting external and internal reproductive structures. The vulva includes external female genital structures. The ovaries are paired organs containing eggs. They produce estrogen and progesterone hormones. Follicles are fluid-filled sacs within ovaries. Each follicle contains one egg. Thousands of follicles exist at birth. This number continuously decreases.
Fallopian tubes connect ovaries to the uterus. Eggs travel through these tubes after ovulation. Fertilization typically occurs in the fallopian tube. The tube’s interior has hair-like projections called cilia. These cilia move the egg toward the uterus. The fimbria are finger-like projections at the tube opening. They capture released eggs. Any damage to tubes impairs fertility significantly.
Male Reproductive Organs
The testes are paired organs producing sperm and testosterone. Seminiferous tubules within testes are where sperm production occurs. This process takes approximately 70 days from start to finish. Sertoli cells support developing sperm. Leydig cells produce testosterone hormone. Both cell types are essential for normal function.
The epididymis is a coiled tube attached to each testis. Sperm mature and gain motility here. Sperm storage also occurs in the epididymis. The vas deferens is a muscular tube transporting sperm. It connects the epididymis to the urethra. Vasectomy cuts or blocks the vas deferens. This prevents sperm from entering ejaculate.
The prostate gland produces fluid contributing to semen. This fluid nourishes and protects sperm. Seminal vesicles produce additional seminal fluid. The bulbourethral glands also contribute secretions. Semen is the complete ejaculated fluid. It contains sperm plus fluids from these glands. The urethra carries semen through the penis during ejaculation. It also carries urine from the bladder.
Hormonal Control of Reproduction
The hypothalamus is a brain region controlling reproduction. It releases gonadotropin-releasing hormone in pulses. This signals the pituitary gland. The pituitary is a small gland at the brain’s base. It produces follicle-stimulating hormone and luteinizing hormone. These hormones travel through blood to reproductive organs. They control egg and sperm production.
Negative feedback regulates hormone levels. Rising sex hormones signal the brain to reduce gonadotropin production. Decreasing sex hormones trigger increased gonadotropin release. This system maintains hormone balance. Disruptions cause fertility problems. Medications can manipulate this system for fertility treatment.
Gametes and Fertilization
Gametes are reproductive cells. Eggs and sperm are gametes. Normal cells contain 46 chromosomes in 23 pairs. Gametes undergo meiosis during development. This special cell division halves the chromosome number. Mature gametes contain 23 single chromosomes. Fertilization combines egg and sperm chromosomes. The resulting embryo has 46 chromosomes again.
Sex chromosomes determine biological sex. Eggs always carry an X chromosome. Sperm carry either X or Y chromosomes. XX combinations produce females. XY combinations produce males. Chromosome abnormalities occur when meiosis errors happen. Extra or missing chromosomes usually prevent normal development. This causes most early pregnancy losses.
Questions about your reproductive anatomy or how specific structures work? Our team explains anatomy in understandable terms. Call +91-8800481100 for detailed explanations.
Fertility Medications and Treatment Drug Terminology
Fertility treatment involves various medications. Understanding drug names and purposes reduces confusion. Most drugs have both generic and brand names. Generic names describe the actual medication. Brand names are specific to manufacturers. Medications fall into several categories based on function and purpose.
Ovulation Induction Medications
Clomiphene citrate is an oral medication stimulating ovulation. Brand names include Clomid and Serophene. The drug blocks estrogen receptors in the brain. The body responds by increasing FSH production. This stimulates follicle development. Typical doses range from 50 to 150 milligrams daily. Treatment lasts five days starting early in the cycle.
Letrozole is an aromatase inhibitor used off-label for fertility. Brand name Femara treats breast cancer at higher doses. For fertility, 2.5 to 5 milligrams daily for five days is standard. Letrozole blocks estrogen production. The pituitary increases FSH similarly to clomiphene. Some evidence suggests letrozole works better than clomiphene for PCOS patients. Side effects are generally minimal.
Injectable Gonadotropins
Injectable FSH medications stimulate multiple follicle development. Follitropin alfa and follitropin beta are recombinant forms. Brand names include Gonal-F and Follistim. These identical medications are produced through genetic engineering. Urofollitropin is purified from postmenopausal women’s urine. Bravelle is one brand name. Human menopausal gonadotropin contains both FSH and LH. Menopur is a common brand.
Dosing varies based on patient age, weight, and ovarian reserve. Starting doses range from 75 to 300 international units daily. Doctors adjust doses based on monitoring results. Injections continue for 8 to 14 days typically. Medications come as powders requiring mixing. Prefilled pens simplify administration. Patients self-administer subcutaneous injections. Proper injection technique training is essential.
Trigger Shot Medications
Human chorionic gonadotropin triggers final egg maturation. This hormone mimics the natural LH surge. Brand names include Pregnyl, Novarel, and Ovidrel. Ovidrel is recombinant hCG requiring no mixing. Others use urinary hCG requiring reconstitution. Dosing ranges from 5,000 to 10,000 international units. Timing must be precise. Egg retrieval or insemination occurs 35 to 36 hours later.
GnRH agonist can also trigger ovulation. Lupron is a commonly used brand. Triggering with agonist reduces OHSS risk significantly. However, luteal phase support becomes more critical. Some protocols use dual trigger. Both hCG and agonist are given together. This may improve egg maturity while reducing complications.
GnRH Antagonists and Agonists
Gonadotropin-releasing hormone antagonists prevent premature ovulation. Ganirelix and cetrotide are commonly used. These medications block GnRH receptors immediately. Daily injections start when follicles reach a certain size. Treatment continues until trigger shot administration. Antagonists don’t require suppression before stimulation begins. This shortens treatment time.
GnRH agonists serve multiple purposes. Initially, they stimulate hormone production. After several days, they suppress the pituitary gland. This suppression prevents premature ovulation. Lupron is the most common agonist. Long protocols use agonist suppression before stimulation. Short protocols add agonist during stimulation. The same medication works differently depending on timing and dosing.
Progesterone Supplementation
Progesterone support is essential after egg retrieval. Natural production is inadequate because retrieval removes progesterone-producing cells. Multiple progesterone formulations exist. Intramuscular progesterone in oil is very effective but requires daily injections. The injection is painful and causes muscle soreness. Subcutaneous progesterone formulations reduce discomfort.
Vaginal progesterone includes suppositories, gels, and tablets. Crinone, Endometrin, and generic forms are available. These avoid injections but can be messy. Oral progesterone is less commonly used. It requires higher doses because of liver metabolism. Combination approaches use multiple formulations together. Progesterone continues through early pregnancy if conception occurs.
Supportive Medications
Estrogen patches or pills prepare the uterine lining. These are essential for frozen embryo transfer cycles. Baby aspirin reduces blood clotting slightly. Some doctors prescribe this to improve implantation. Antibiotics prevent infection after egg retrieval. Doxycycline is commonly prescribed. Prenatal vitamins should begin before conception. Folic acid prevents neural tube defects. Additional supplements may be recommended based on individual needs.
Overwhelmed by Medication Instructions?
We understand fertility medications can feel overwhelming. Our nursing staff provides detailed medication teaching and remains available throughout your treatment cycle to answer questions.
Laboratory Values, Results, and Testing Terminology
Laboratory testing provides crucial fertility information. Understanding test results helps patients participate in treatment decisions. Numbers alone don’t tell the complete story. Context matters. Age affects interpretation of many values. Lab ranges represent averages. Individual results require personalized assessment. Doctors consider multiple factors when interpreting results.
Hormone Level Interpretation
FSH measured on cycle day two or three assesses ovarian reserve. Normal values vary by laboratory. Generally, FSH under 10 IU/L is considered normal. Values between 10 and 15 suggest diminishing reserve. FSH exceeding 15 indicates significant reserve decline. Very high FSH over 20 means poor reserve. Treatment response will likely be minimal. However, FSH fluctuates between cycles. One elevated value doesn’t define prognosis definitively.
Anti-Müllerian hormone provides additional reserve information. This value remains stable throughout the menstrual cycle. Higher AMH indicates more remaining follicles. AMH over 2.0 nanograms per milliliter suggests good reserve. Values between 1.0 and 2.0 are adequate. AMH under 1.0 indicates diminished reserve. Very low AMH below 0.5 suggests poor stimulation response. Extremely high AMH may indicate PCOS.
Estradiol on day two or three should be low. Elevated baseline estradiol suggests a cyst or premature follicle development. This may affect cycle timing. During stimulation, rising estradiol confirms follicle growth. Each mature follicle produces approximately 200 to 300 picograms per milliliter. Very high estradiol increases OHSS risk. Doctors may adjust medication or delay trigger if estradiol rises too quickly.
Progesterone Testing
Progesterone confirms ovulation occurred. Testing happens approximately one week after suspected ovulation. This coincides with mid-luteal phase. Progesterone over 3 nanograms per milliliter confirms ovulation. Values over 10 indicate strong ovulation. Very low progesterone suggests anovulation or poor corpus luteum function. During IVF cycles, progesterone supplementation makes this test unhelpful. Exogenous progesterone raises levels regardless of natural production.
Pregnancy Testing
Quantitative hCG measures exact hormone levels. This blood test is more sensitive than home pregnancy tests. Values over 5 mIU/mL indicate pregnancy. In early pregnancy, hCG should double every 48 to 72 hours. Slow rises suggest problems. Very high hCG may indicate multiple pregnancies or molar pregnancy. Serial measurements track pregnancy progression. Declining hCG indicates pregnancy loss.
Qualitative pregnancy tests give yes or no results. Home pregnancy tests work this way. Blood qualitative tests are rarely necessary. Most clinics use quantitative testing. The first hCG test occurs about two weeks after embryo transfer. This is called beta day. A second test follows 48 hours later. The doubling pattern predicts viability better than single values.
Semen Analysis Parameters
Volume measures the ejaculated semen quantity. Normal range is 1.5 to 5 milliliters. Low volume suggests collection problems or anatomical issues. Very high volume dilutes sperm concentration. Concentration counts sperm per milliliter. Normal concentration exceeds 15 million per milliliter. Total count multiplies concentration by volume. At least 39 million total sperm should be present.
Motility percentages indicate moving sperm. Progressive motility specifically counts forward-moving sperm. At least 32% should show progressive movement. Total motility includes all movement types. Morphology assesses sperm shape. By strict Kruger criteria, at least 4% should have normal form. Some labs use WHO criteria where 15% normal is acceptable. Additional tests evaluate vitality, DNA fragmentation, and antibodies when indicated.
Ultrasound Measurements
Antral follicle count is performed during baseline ultrasound. Small follicles measuring 2 to 10 millimeters are counted on both ovaries. Total count predicts ovarian reserve and stimulation response. Count over 12 suggests good reserve. Count between 6 and 12 is adequate. Counts under 6 indicate poor reserve. Very high counts over 25 may indicate PCOS.
During stimulation, growing follicles are measured. Mature follicles typically measure 16 to 22 millimeters. Lead follicles are the largest ones. When multiple follicles reach maturity, trigger timing is determined. Endometrial thickness is also measured. Adequate thickness is at least 7 millimeters. Thicker linings over 8 to 10 millimeters are better. Pattern also matters. Trilaminar appearance is ideal.
Confused about your test results? Our specialists interpret your numbers and explain what they mean for your fertility treatment. Call +91-8800481100 to discuss your results.
Understanding Fertility Treatment Success Rates and Statistics
Success rates help patients understand treatment effectiveness. However, statistics can be misleading without context. Multiple ways exist to calculate and report success. Understanding these differences prevents unrealistic expectations. Individual results vary significantly from published averages. Personal factors affect outcomes more than general statistics suggest.
Pregnancy Rate Versus Live Birth Rate
Pregnancy rate measures positive pregnancy tests. This includes all pregnancies regardless of outcome. Some pregnancies end in miscarriage. Chemical pregnancies count toward pregnancy rates. Clinical pregnancy rate requires ultrasound confirmation of gestational sac. This provides more meaningful information than biochemical pregnancy rates alone.
Live birth rate represents actual babies born. This is the most important statistic for patients. Live birth rate per cycle started counts all attempts. This includes cycles canceled before egg retrieval. Live birth rate per embryo transfer excludes canceled cycles. This inflates apparent success. Cumulative live birth rate across multiple cycles provides realistic expectations. Most live births occur within three to four IVF attempts.
Age and Success Rates
Maternal age dramatically affects all fertility treatment outcomes. Women under 35 have highest success rates. Pregnancy and live birth rates decrease gradually from 35 to 40. Decline accelerates after 40. By age 44, success rates drop below 5% per cycle in most cases. Donor eggs eliminate age effects on success rates. The egg donor’s age matters while recipient age matters much less.
Male age also influences outcomes though less dramatically. Advanced paternal age increases miscarriage risk. Sperm DNA damage accumulates with age. However, men maintain fertility potential much longer than women. Combined parental age matters when both partners are older. Success rates reflect the more significant limiting factor.
Singleton Versus Multiple Birth Rates
Multiple pregnancy rates vary with embryo number transferred. Single embryo transfer dramatically reduces twin risk. Some embryos split naturally creating identical twins. This occurs in about 1% to 2% of IVF pregnancies. Transferring two embryos increases twin chances significantly. Both embryos may implant. One might split creating triplets from two embryos.
Multiple pregnancies carry higher risks. Preterm birth is much more common. Low birth weight affects most multiples. Pregnancy complications increase. Mother’s health risks rise substantially. Neonatal intensive care is often necessary. Long-term developmental issues are more frequent. These risks explain the push toward single embryo transfer.
Miscarriage Rates
Miscarriage risk increases with maternal age. Women under 35 have approximately 15% miscarriage risk. This rises to 25% by age 40. After 42, miscarriage exceeds 50% of pregnancies. Chromosome abnormalities cause most age-related losses. Genetic testing of embryos may reduce miscarriage rates. However, testing doesn’t eliminate all loss. Other factors contribute beyond chromosomes.
Clinic-Specific Versus National Statistics
Individual clinics report their own success rates. These vary considerably between programs. Patient population differences partly explain variations. Clinics accepting harder cases may show lower success rates. However, they might actually provide better care. Cherry-picking easy cases inflates statistics artificially. National databases compile all clinic data. This provides better overall picture of expected success.
The Society for Assisted Reproductive Technology maintains U.S. data. Centers for Disease Control also publishes fertility clinic success rates. These reports lag two years behind current cycles. Treatment protocols evolve continuously. Historical data may not reflect current practices. Individual consultation provides more accurate personal predictions than published statistics.
Want to Know Your Personal Success Probability?
General statistics don’t tell your individual story. We assess your specific situation and provide personalized success predictions based on your unique circumstances.
Moving Forward: Your Next Steps in Understanding Fertility
Understanding infertility terminology empowers you throughout your fertility journey. Medical language no longer needs to feel intimidating. You now have knowledge to communicate effectively with your healthcare team. This comprehensive glossary covered hundreds of terms across all aspects of fertility care. From basic anatomy to complex procedures, you’ve gained essential vocabulary.
Knowledge transforms anxiety into confidence. When doctors discuss your treatment plan, you’ll understand the reasoning. Test results make sense in context. Medication purposes become clear. Procedure steps are no longer mysterious. This understanding helps you participate actively in decisions affecting your reproductive future.
Remember that every fertility journey is unique. Your specific situation requires personalized evaluation and treatment. While this glossary provides general information, individual circumstances vary widely. What works for one person may not work for another. Age, diagnosis, and personal factors all influence appropriate treatment approaches.

Author Bio: Neelam Chhagani is an International Surrogacy Expert with 15 years of experience in the fertility and surrogacy domain. As the founder of IVF Conceptions and Complete Surrogacy, she has guided over 4,000 intended parents worldwide on their surrogacy journey to parenthood. Recognized as a trusted authority, she specializes in holistic infertility solutions and third-party reproduction consulting.
Holding an MA in Counselling Psychology and a PGD in Mental Health, Neelam is a proud member of the European Fertility Society (EFS) and the European Society of Human Reproduction and Embryology (ESHRE). She is also a leading surrogacy blogger, providing valuable insights into ethical and practical surrogacy solutions.
Since 2010, committed to supporting ALL family types, Neelam has been passionate about helping intended parents grow their families with compassion, integrity, and a focus on secure and affordable surrogacy options Globally.
Learn more about Neelam:
https://www.ivfconceptions.com/neelam-chhagani-surrogacy-consultant/
https://www.linkedin.com/in/neelam-chhagani-92892229/


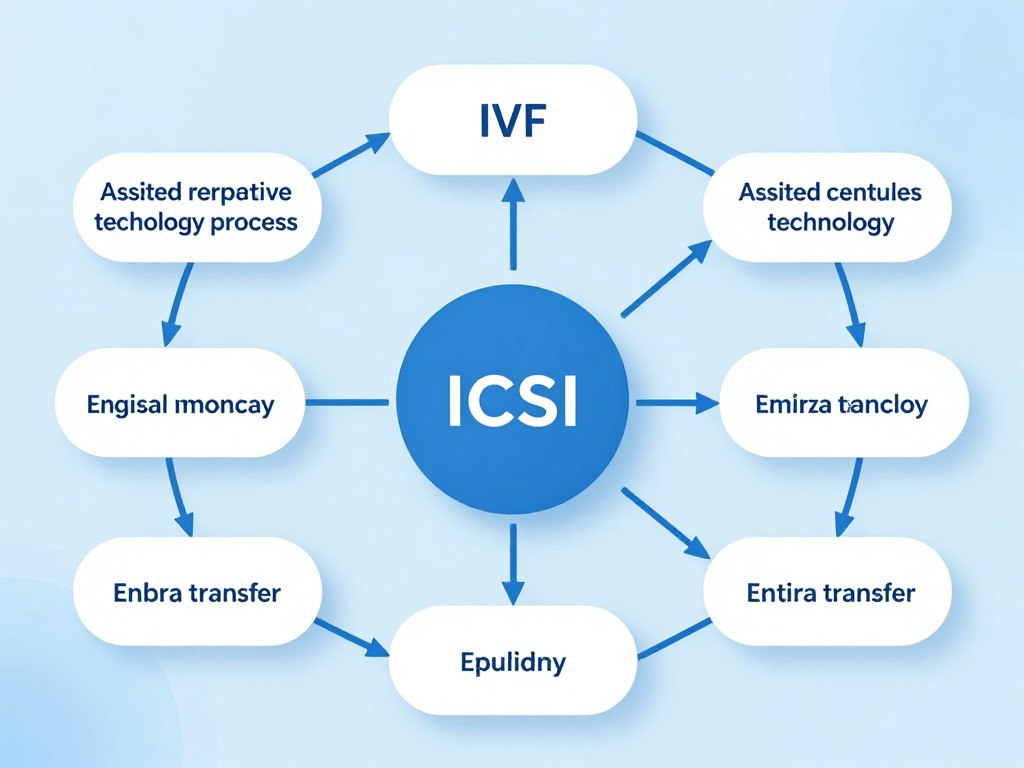

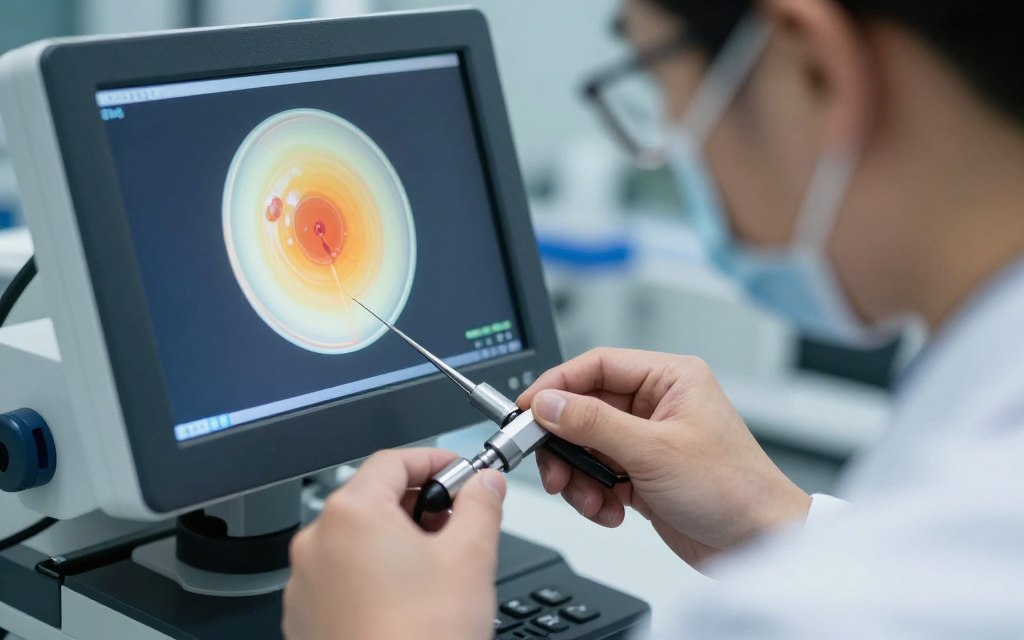
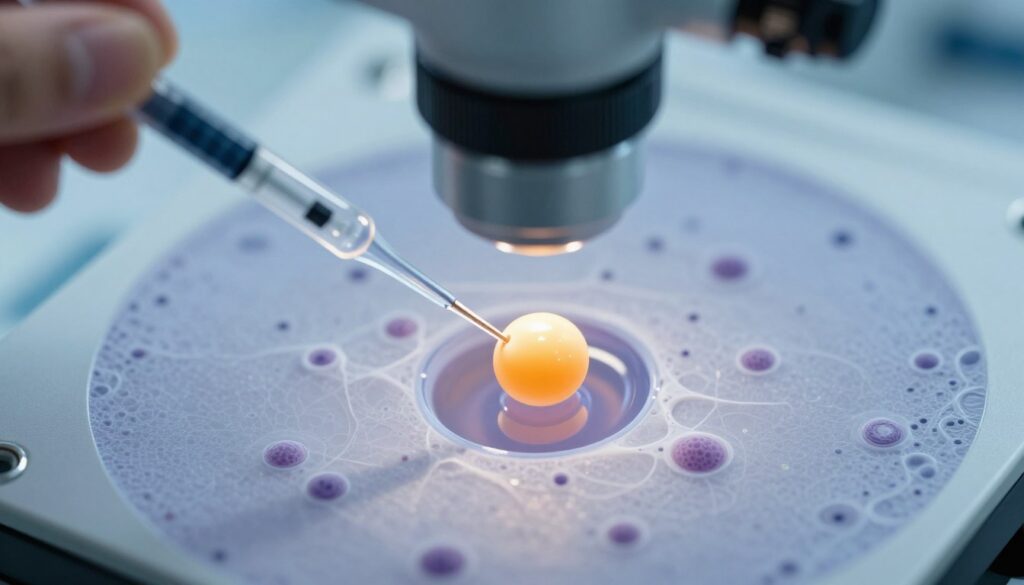
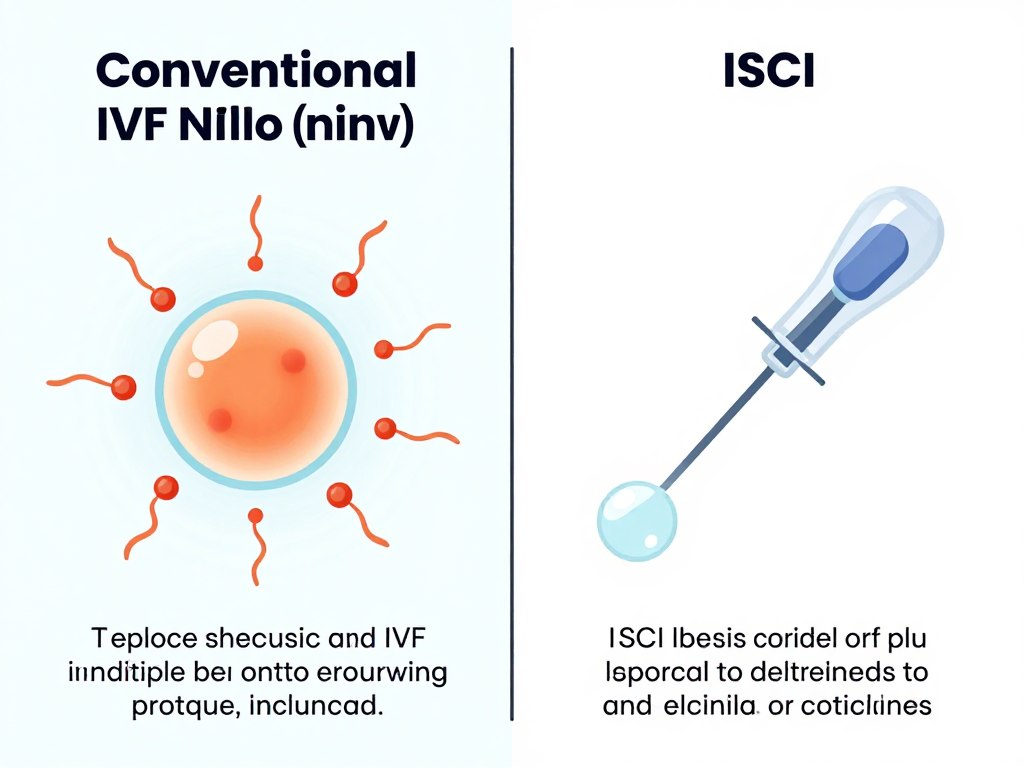
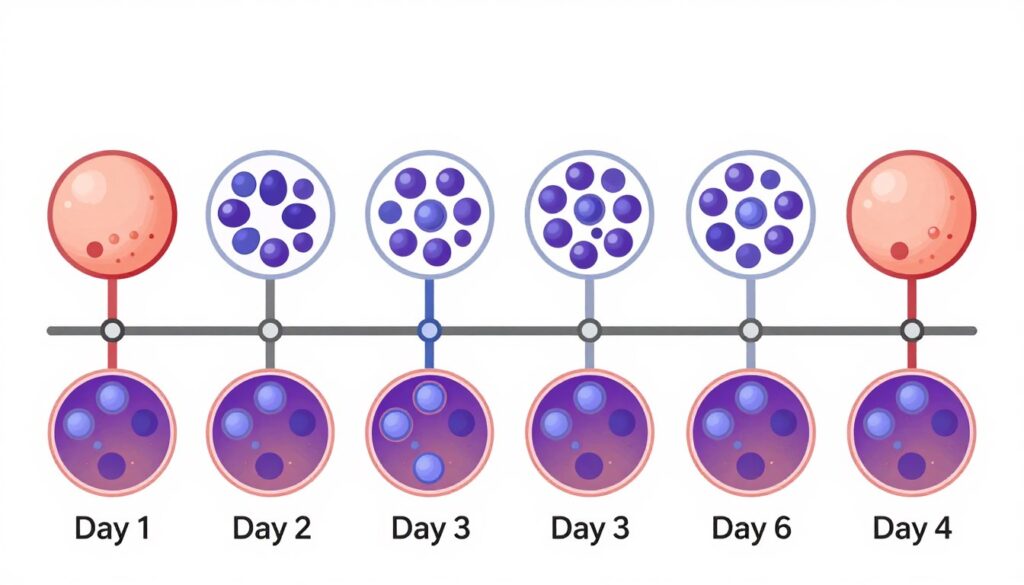

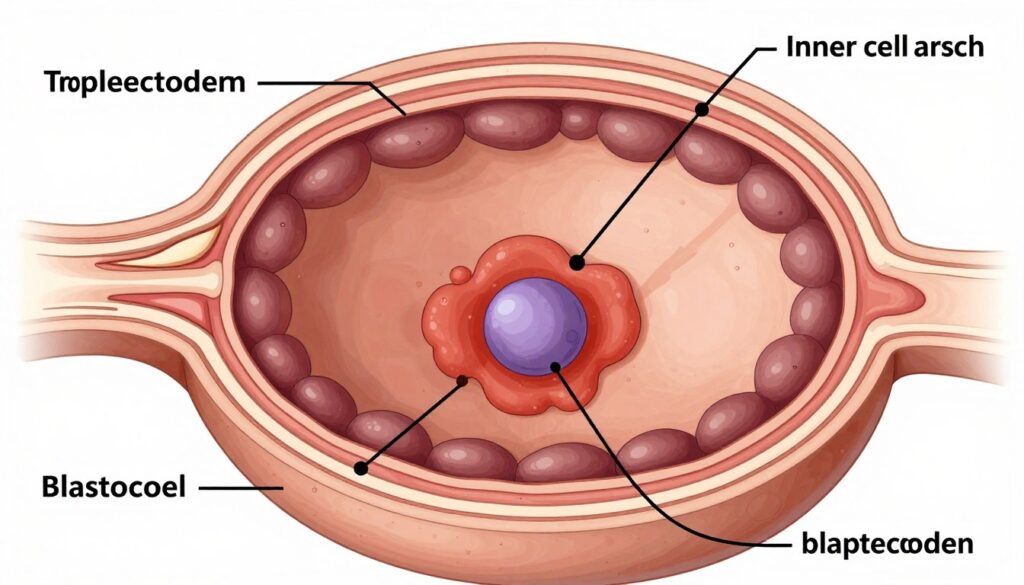
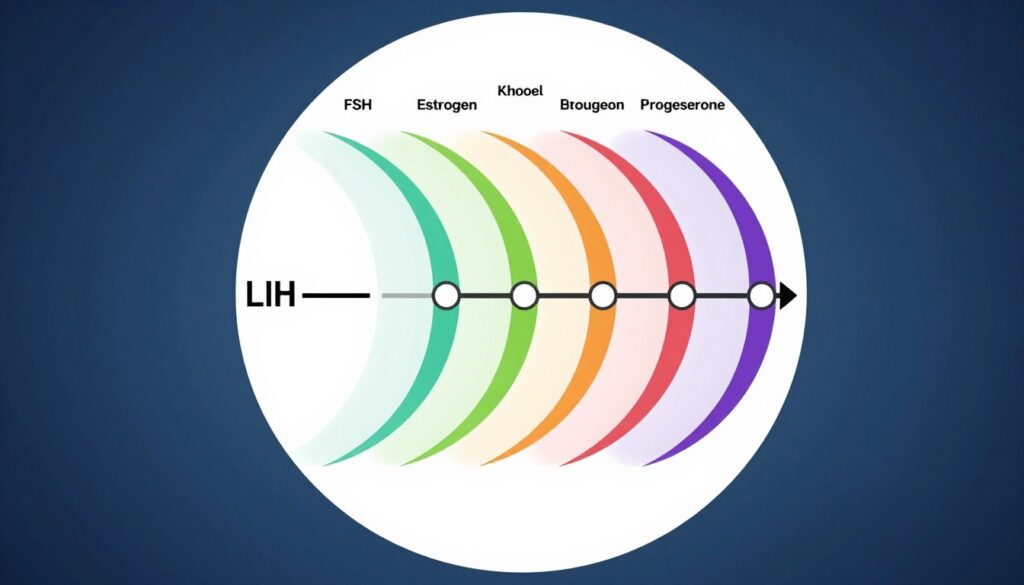

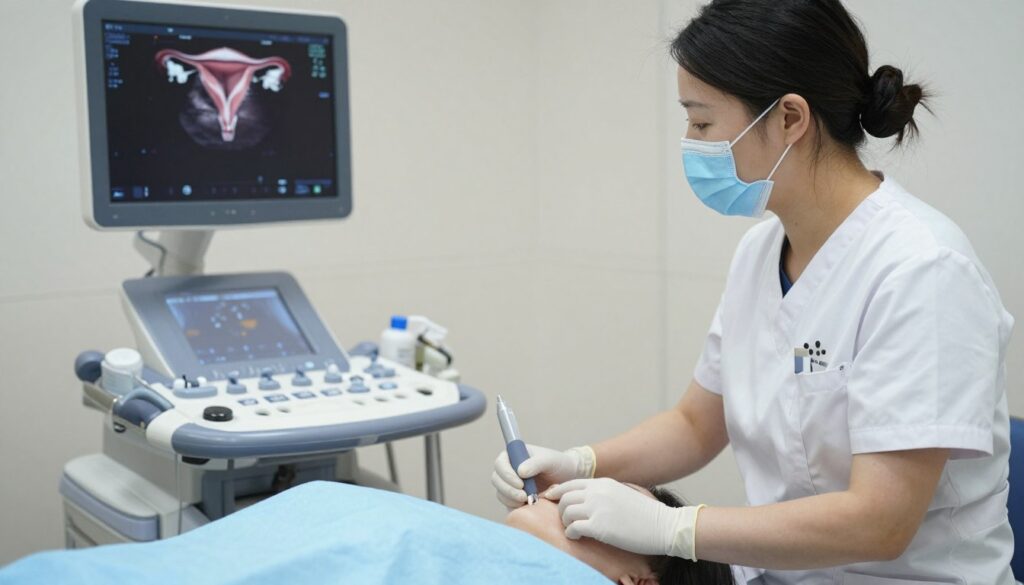
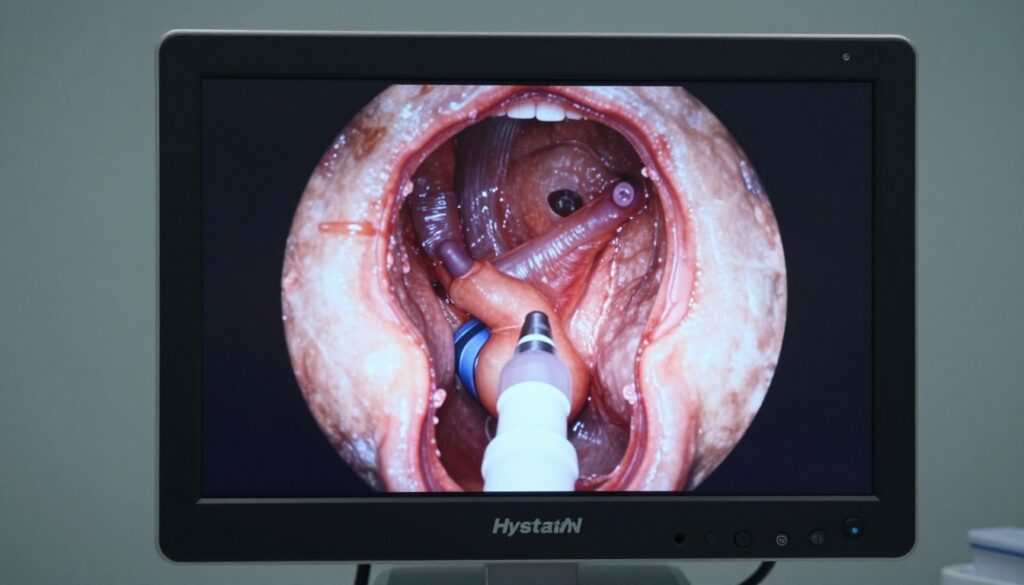
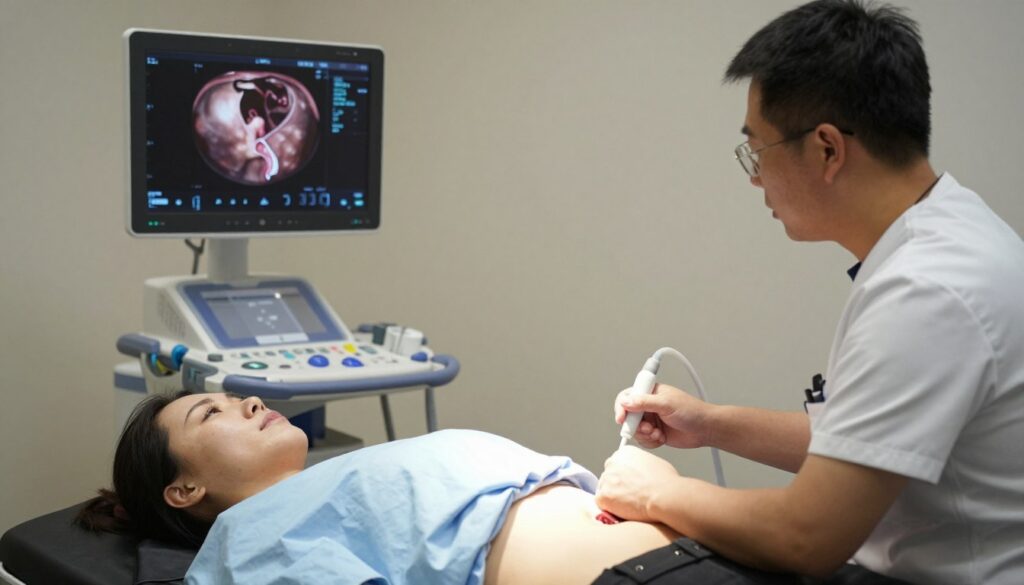


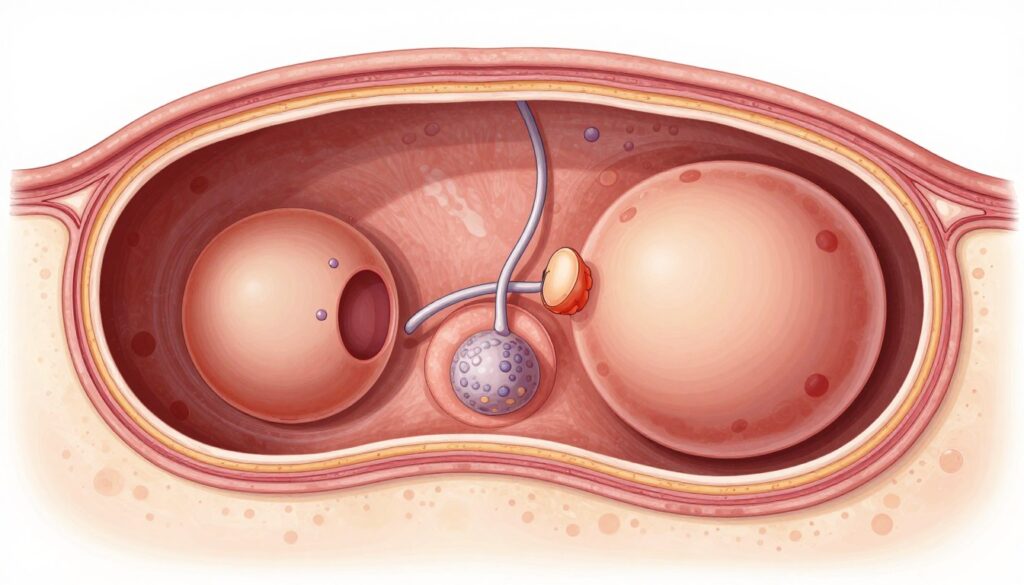

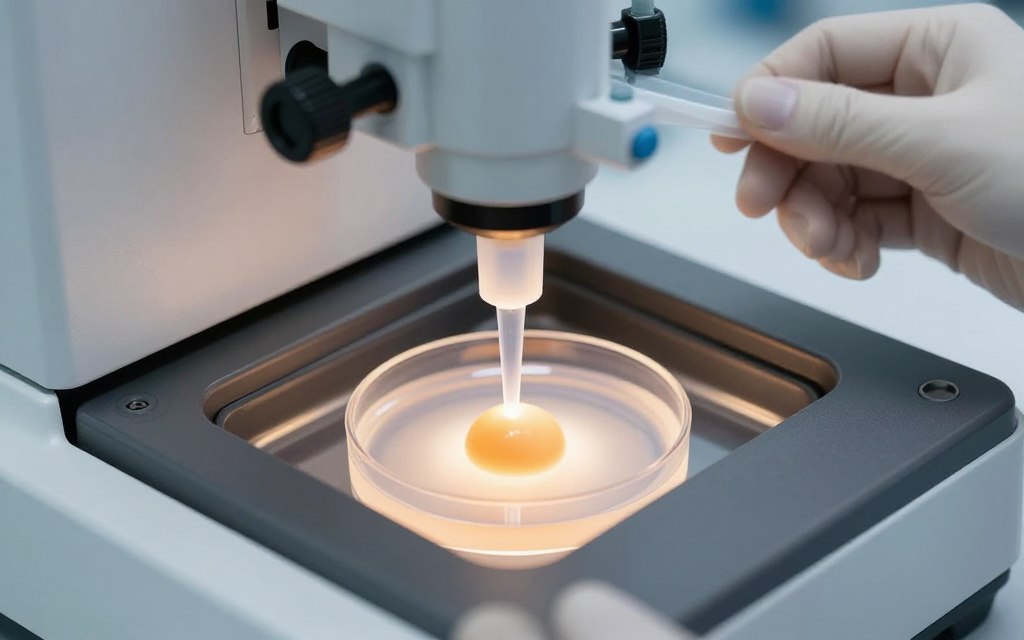


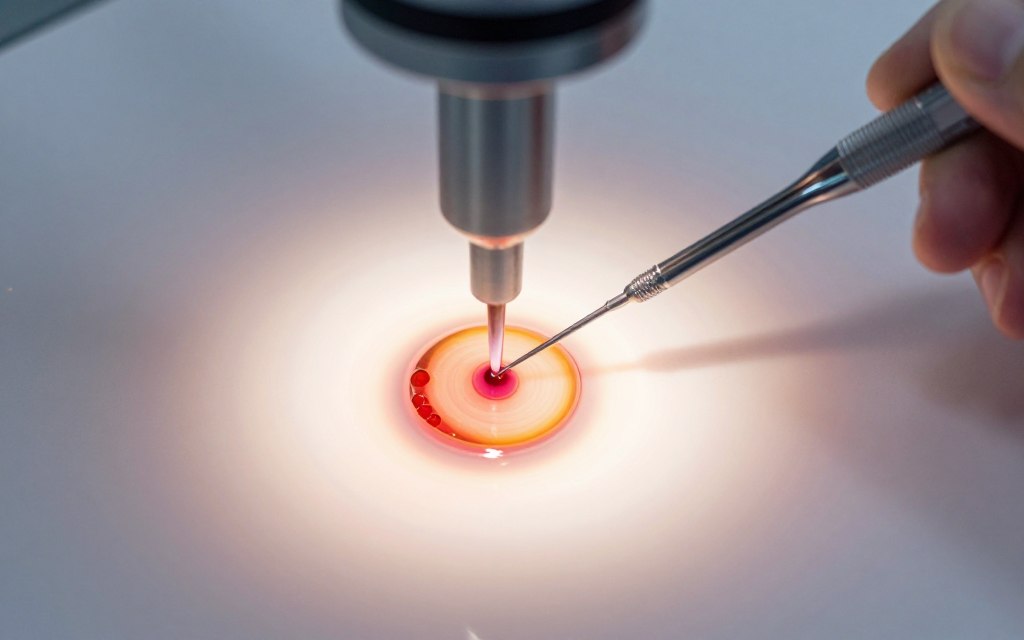
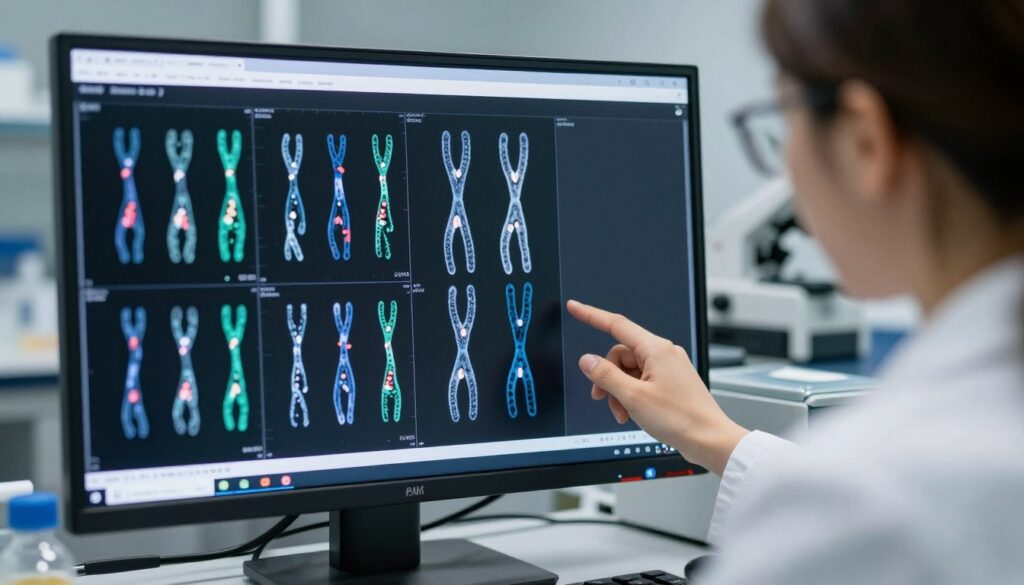




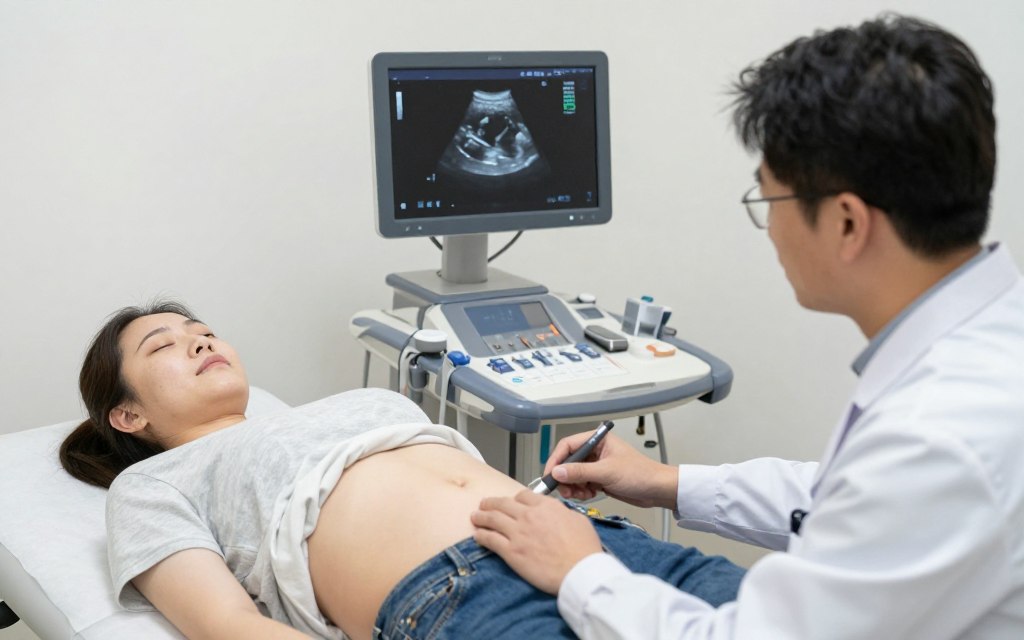




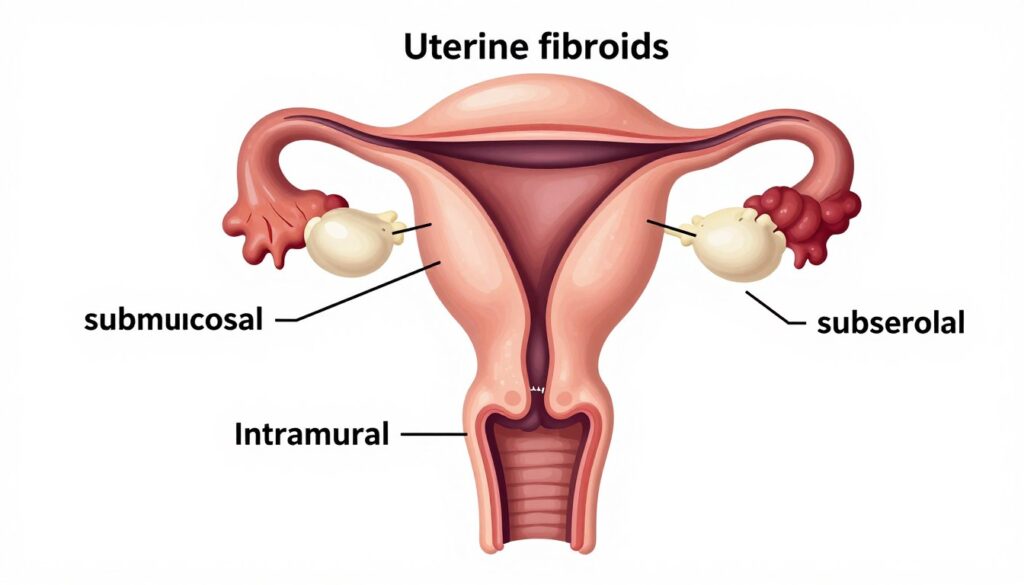


















I was introduced to Neelam by a friend who worked with Neelam for surrogacy. Neelam is absolutely wonderful. I am a single male and the journey to fatherhood is not that easy. Neelam connected me to a program ideal for my circumstances. She was with me throughout the pregnancy providing advice and guidance along the way. I am so grateful I found her and am thrilled today that I have a beautiful daughter. I highly recommend Neelam to anyone who is on a journey to become a parent. Having a child has changed my world for the better. I wish others success with their own journey and recommend you connect with Neelam to find a path that is best for you.
SA (USA)